Analysis of chromium distribution in tissues of bottlenose, striped and Risso’s dolphins from the Croatian Adriatic over a 20-year period
M.Đokić, N. Bilandžić, M. Benić, T. Bolanča, T. Gomerčić, M. Đuras and M. Sedak*
Maja ĐOKIĆ1, dokic@veinst.hr, orcid.org/0000-0002-3071-6208; Nina BILANDŽIĆ1, bilandzic@veinst.hr, orcid.org/0000-0002-0009-5367;
Miroslav BENIĆ2, benic@veinst.hr, orcid.org/0000-0001-7594-520X; Tomislav Bolanča3, tbolanca@fkit.unizg.hr, orcid.org/0000-0002-2268-
8139; Tomislav GOMERČIĆ4, tomislav.gomercic@vef.unizg.hr, orcid.org/0000-0001-7232-9428; Martina ĐURAS5, martina.duras@vef.hr, orcid.
org/0000-0002-0538-0992; Marija SEDAK1* (corresponding author), sedak@veinst.hr, orcid.org/0000-0001-6861-0436.
1 Laboratory for the Deterimation of Residues, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
2 Laboratory for Mastitis and Row Milk Quality, Department of Bacteriology and Parasitology, Croatian Veterinary Institute, 10000 Zagreb, Croatia
3 Department of Analytical Chemistry, Faculty of Chemical Engineering and Technology, University of Zagreb, 10000 Zagreb, Croatia
4 Department of Veterinary Biology, Faculty of Veterinary Medicine, University of Zagreb, 10000 Zagreb, Croatia
5 Department of Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, University of Zagreb, 10000 Zagreb, Croatia
https://doi.org/10.46419/cvj.57.4.1
Abstract
This study aimed to quantify chromium (Cr) concentrations in tissues of 190 individuals of three odontocete species—159 bottlenose dolphin (Tursiops truncatus), 25 striped dolphin (Stenella coeruleoalba), and six Risso’s dolphin (Grampus griseus)—stranded along the Croatian coast of the Adriatic Sea between 1995 and 2013. Chromium concentrations were measured in liver, muscle, kidney, skin, lung, spleen, and fat using inductively coupled plasma optical emission spectrometry. Significant interspecific differences in Cr concentrations were observed in the kidney, liver, and spleen, suggesting dietary habits as a major factor in chromium accumulation. Risso’s dolphins exhibited the highest Cr levels, likely due to the higher Cr concentrations in cephalopods that make up the basis of their diet. Chromium concentrations exceeding 1 mg/kg wet weight were found in only 16 tissue samples—15 from bottlenose dolphin and one from striped dolphin —indicating a low overall prevalence of elevated Cr levels. Regression analysis revealed no significant correlation between Cr concentrations and body mass, age, sex, or geographic location. However, a significant decline in Cr levels over the study period was observed across all tissues.
Key words: chromium, Tursiops truncatus, Stenella coeruleoalba, Grampus griseus, tissues, Adriatic Sea
Introduction
Chromium (Cr) in the marine environment originates from both natural and anthropogenic sources. The predominant natural input is associated with continental dust (ATSDR, 2012). However, human activities contribute a significantly larger proportion of environmental chromium, primarily through the combustion of fossil fuels (gas, coal, and oil), emissions from metal industries, and wastewater discharges from electroplating, leather processing, and textile manufacturing (ATSDR, 2012). While the sources of chromium are well established, its fate, transport, and speciation in marine environments remain insufficiently understood.
In seawater, hexavalent chromium (Cr⁶⁺) is the predominant form, with an estimated residence time ranging from 4.6 to 18 years (Pettine and Millero, 1990; ATSDR, 2012). Total dissolved Cr⁶⁺ concentrations range from 1.2 nmol/L in unpolluted waters to 365 nmol/L in areas affected by industrial wastewater discharges. Chromium exists primarily in two oxidation states in marine systems: trivalent (Cr³⁺) and hexavalent (Cr⁶⁺). The relative abundance of these forms is governed by redox conditions, with Cr⁶⁺ as the dominant species, typically as chromate (CrO₄²+). In the Mediterranean Sea, chromate concentrations range between 3 and 4 nmol/L, while Cr³⁺ levels generally remain below 1 nmol/L. Key anthropogenic sources of chromium include the electroplating, leather, paint, and wood preservation industries. The average enrichment factor for Cr in sediments of coastal areas and lakes is 2.2, and the anthropogenic mobilisation factor is 1.6. Chromium poses toxicity risks to aquatic organisms, with acute toxicity observed in fish at concentrations exceeding 10 mg/L and sublethal effects detected in phytoplankton and fish at concentrations as low as 10 µg/L (Bjerregaard et al., 2015).
Despite its environmental prevalence and toxicity, few scientific studies have focused on chromium pollution in marine ecosystems, particularly concerning its speciation and impact on marine mammals. One major limitation is the biological reduction of Cr⁶⁺ to Cr³⁺, which hinders direct quantification of the more toxic species. Consequently, most available data refer to total Cr concentrations, particularly in large marine mammals such as whales, where tissue levels are typically very low, generally under 1 mg/kg (Wise et al., 2009).
In mammals, the most significant toxic effects are associated with Cr⁶⁺, which is approximately 100 times more toxic than Cr³⁺. Cr⁶⁺ is absorbed more efficiently and accumulates primarily in red blood cells (Eisler, 2010). However, in the mammalian digestive system, gastric acid reduces Cr⁶⁺ to Cr³⁺, which has a gastrointestinal absorption rate of less than 1% (Förstner and Wittmann, 1983). While mammals can typically tolerate chromium concentrations 100–200 times above normal without adverse effects, chronic exposure has been linked to dysfunction of the hypothalamic-pituitary axis, central nervous system, and reproductive system, along with hepatic, renal, and cardiovascular damage (Yasutake and Hirayama, 2002). Notably, no correlation has been observed between chromium levels and age in marine mammals (Eisler, 2010). For instance, no alteration in lymphocyte proliferation was detected in newborn harbour seals (Phoca vitulina ) exposed to 5.0 mg/L of chromium for five days (Eisler, 2010). Although chromium plays a biological role in carbohydrate and lipid metabolism (Tokar et al., 2013), research on its accumulation in marine mammal tissues remains limited. This lack of data represents a notable gap in environmental toxicology, particularly given the potential for Cr to exert harmful effects at elevated concentrations. Comprehensive assessment of Cr distribution in marine mammals is therefore essential for understanding both ecological risks and species-specific exposure levels.
The Adriatic Sea, a subregion of the Mediterranean basin, hosts a resident population of bottlenose dolphins (Tursiops truncatus), with occasional sightings of other cetacean species (Pompe-Gotal et al., 2009). However, data on trace element concentrations in dolphins from the Croatian Adriatic are still relatively scarce. Most existing studies have predominantly addressed non-essential trace elements in tissues of stranded bottlenose and striped dolphins, as well as broader evaluations of toxic element loads in these and in Risso’s dolphins (Grampus griseus) (Bilandžić et al., 2012, 2015; Šuran et al., 2015; Sedak et al., 2022, 2025). To date, only three studies have focused on the concentrations of essential trace elements in the tissues of these cetacean species (Bilandžić et al., 2016; Đokić et al., 2018, 2025).
The primary objective of this study was to assess Cr concentrations in various tissues, including muscle, liver, kidney, lung, spleen, adipose tissue, and skin of bottlenose, striped, and Risso’s dolphins stranded along the Croatian coast of the Adriatic Sea between 1995 and 2013. These cetacean species are widely recognised as sentinel organisms for monitoring marine pollution. This research aims to enhance the current understanding of the contamination status of these species and to contribute valuable data on Cr levels in the Adriatic Sea, particularly within Croatian waters.
Material and methods
Samples
Tissue samples were collected from dolphins stranded along the Croatian Adriatic coast between 1995 and 2013. A total of 190 individuals were included in the study: 159 bottlenose dolphins (Tursiops truncatus), 25 striped dolphins (Stenella coeruleoalba), and six Risso’s dolphins (Grampus griseus).
Mean body length (cm, mean ± SD) for each species was: bottlenose dolphin 239.2 ± 53.9, striped dolphin 191.5 ± 40.3, and Risso’s dolphin 300.1 ± 10.6. Corresponding mean body weight (kg, mean ± SD) was: bottlenose 150.5 ± 79.8, striped 81.1 ± 15.4, and Risso’s 258.8 ± 46.4.
As part of the post-mortem examination, a range of biological and morphometric data were recorded, including species, sex, body mass, total body length, and other standard external measurements. Prior to dissection, age, sex, body weight, and length were recorded. During necropsy, teeth were collected for age estimation. Tooth sections were prepared following the methodology of Slooten (1991), and age was determined by counting growth layer groups according to Hohn et al. (1998). Individuals older than seven years were classified as adults, based on criteria established by André et al. (1991).
Among bottlenose dolphins, 77 were males and 81 females. Age data identified 77 adults and 52 juveniles. Age could not be determined for 30 individuals and sex for one individual. Striped dolphins were categorised solely by sex (15 males and 10 females) due to missing age data. All six Risso’s dolphins were adults; however, sex was not determined due to the small sample size.
For spatial analysis, the Croatian Adriatic Sea was divided into the north and south geographic regions, with a higher proportion of samples originating from the south region. Following sex and body length determination, tissue samples were excised, sealed in plastic bags, and stored at -20°C until further analysis.
Sample Preparation and Elemental Analysis
Tissue samples (0.5 g) were subjected to microwave-assisted digestion using Teflon vessels. The digestion mixture consisted of 2.5 mL nitric acid (65%), 1.0 mL hydrogen peroxide (30%) (Suprapur® grade; Merck KGaA, Darmstadt, Hesse, Germany), and 3.0 mL ultrapure water. After digestion and subsequent cooling, an internal standard, Yttrium (Y), was added to each sample to a final concentration of 200 μg/L, followed by dilution to a final volume of 50 mL with ultrapure water in a volumetric flask.
Elemental concentrations were expressed in milligrams per kilogram (mg/kg) on a wet weight basis. For comparison with values reported on a dry weight basis in the literature, concentrations were converted using a standard wet-to-dry weight conversion factor of 0.25, as recommended by Becker et al. (1995).
Cr quantification was performed using an inductively coupled plasma optical emission spectrometer (ICP-OES), Model Optima 8000, equipped with an S10 autosampler (PerkinElmer, Waltham, MA, USA). Calibration was performed using multi-element Environmental Standards for ICP-MS (1000 mg/L, ISO 17034 certified, Agilent Technologies, Santa Clara, CA, USA). The concentration range of the Cr calibration curve was from 1 to 100 μg/L. Recalibration using the standard solution at the midpoint concentration of the calibration curve was performed every 20 measurements to ensure instrument stability and accuracy. Quality assurance and quality control (QA/QC) protocols included the analysis of procedural blanks, replicate samples, and certified reference materials (CRMs), specifically DORM-4 and DOLT-5, obtained from the National Research Council (NRC), Institute for National Measurement Standards (Ottawa, ON, Canada). Recovery rates for CRMs and instrumental quantification limits for each element are detailed in Table 1.
Table 1. Analytical recovery of chromium in certified reference materials (CRM), accompanied by the corresponding instrumental limits of detection (LOD) and quantification (LOQ).

Statistical Analysis
All statistical analyses were conducted using Stata version 13.1 (StataCorp, College Station, TX, USA). For each trace element, descriptive statistics including the mean, standard deviation, median, minimum, and maximum were calculated. The Shapiro–Wilk test was applied to evaluate the normality of the distribution of element concentrations across tissue types.
Comparisons of element concentrations among the three dolphin species were performed using the non-parametric Kruskal-Wallis test. Statistical significance was assessed using p-values.
To explore linear relationships between the logarithm of element concentrations and independent variables (body mass, body length, age, sex, sampling location, and year of discovery), linear regression analysis was performed. The regression model took the form:
log c = log b × x + a,
where c represents the mass concentration of the element in the tissue [mg/kg], a is the intercept, log b is the slope (regression coefficient), and x is the independent variable.
The regression results are presented in terms of the antilogarithmic values of the regression coefficients, denoted as b. These values indicate the magnitude of change in element concentration associated with a one-unit change in the independent variable. A value of b greater than 1 implies an increase in concentration, while a value less than 1 indicates a decrease.
Spearman’s rank correlation coefficient was used to evaluate relationships between element concentrations within the same tissue type across individuals, as well as between different tissues within the same individual. The strength of these correlations was interpreted using a commonly accepted scale:
- 00 to 0.19 — very weak
- 20 to 0.39 — weak
- 40 to 0.59 — moderate
- 60 to 0.79 — strong
- 80 to 1.00 — very strong
Results and discussion
Cr concentrations in the tissues of three dolphin species stranded along the Croatian coast of the Adriatic Sea between 1995 and 2013 are presented in Table 2. Values are expressed as the mean ± standard deviation (SD), median and ranges on a wet weight basis.
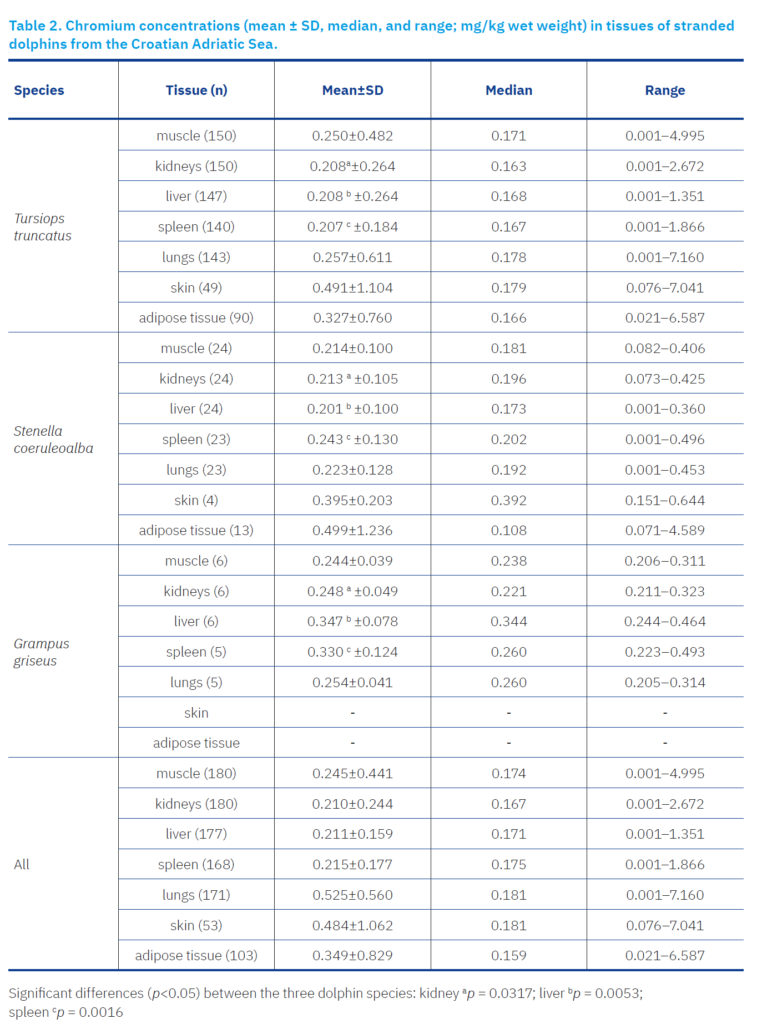
In most analysed samples, Cr concentrations in the tissues of all three dolphin species were approximately 0.2 mg/kg. However, significant variations were observed among species, tissues, and individual animals.
In the bottlenose dolphin, the lowest Cr concentrations were recorded in the skin of an adult male (mean 0.12 mg/kg; median 0.13 mg/kg), while the highest concentrations were found in the lungs of a 13-year-old female (7.16 mg/kg). Exceptionally high values were measured in the blubber and muscle tissues of certain individuals (up to 6.59 mg/kg and 4.99 mg/kg, respectively), which significantly influenced the mean values, whereas the medians remained much lower, indicating an asymmetric distribution of the data (Figure 1).
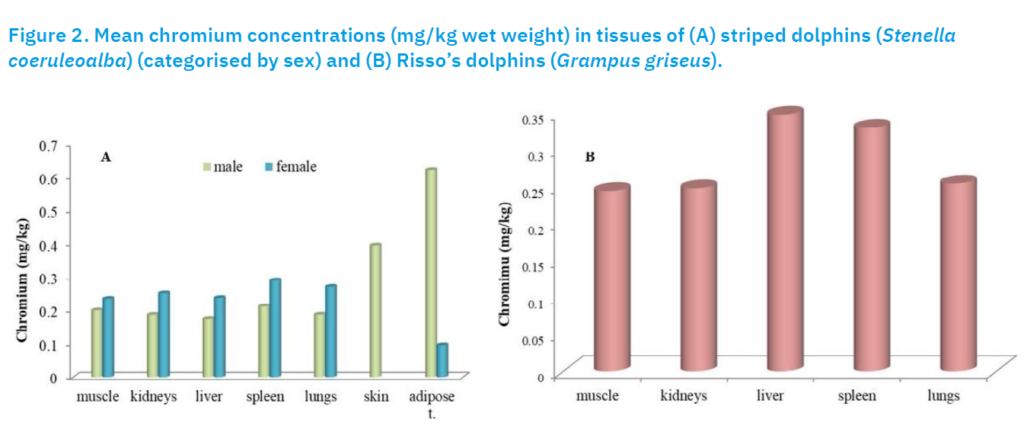
In Risso’s dolphin, Cr concentrations were more uniform, with smaller differences between the mean values and medians. The highest values were recorded in the liver (0.35 mg/kg) and spleen (0.33 mg/kg), which were also higher compared to the other species.
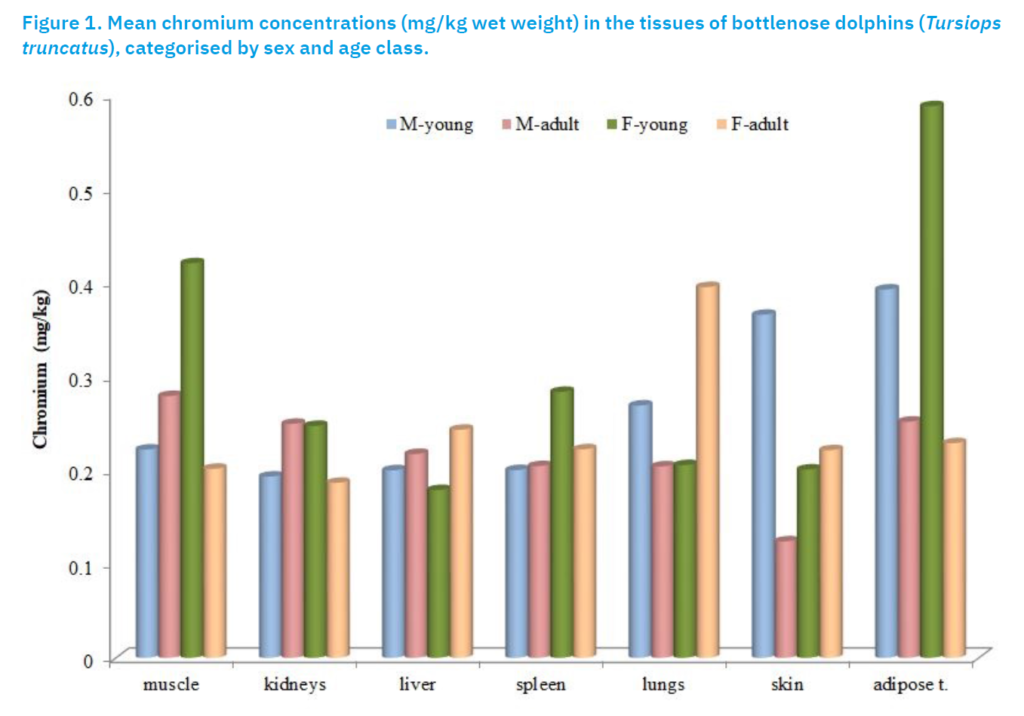
In striped dolphins, Cr concentrations were generally higher in females than in males, with the exception of blubber tissue, where data for females were limited (n = 3). One extreme value (4.59 mg/kg) in the blubber of an 18-year-old male significantly increased the mean value in that group (Figure 2).
Statistically significant interspecific differences in Cr concentrations were found in the kidney (p = 0.0317), liver (p = 0.0053), and spleen (p = 0.0016). The highest mean hepatic Cr concentration was recorded in Risso’s dolphin (0.35 mg/kg), while lower but similar levels were found in striped (0.20 mg/kg) and bottlenose dolphin (0.21 mg/kg). These results support the role of diet as a key factor in trace element accumulation.
Diet is the main route of chromium exposure in mammals. Chromium (VI) compounds are absorbed more efficiently (2–10%) than chromium (III) compounds (0.5–3%) (Outridge & Scheuhammer, 1993). Although data are limited, cephalopods generally contain more Cr than fish (Hansen et al., 2016), suggesting that species-specific feeding habits explain the observed differences. Risso’s dolphin feeds mainly on squid (~85%), striped dolphin has a mixed diet, while bottlenose dolphin primarily consumes fish (~60%) with minimal squid intake (Pauly et al., 1998).
In uncontaminated habitats, tissue Cr concentrations typically range from 0.025 to 3.75 mg/kg wet weight, depending on tissue type. In polluted areas, levels may be up to two orders of magnitude higher (Outridge & Scheuhammer, 1993), highlighting the combined impact of environmental exposure and trophic ecology on Cr accumulation.
In this study, Cr concentrations exceeding 1 mg/kg wet weight were detected in only 16 tissue samples, representing only a small proportion of the total. Of these, 15 originated from bottlenose dolphin and one from striped dolphin. According to Eisler (1986), Cr concentrations above this threshold may suggest environmental contamination. In animals with chronic exposure, bone tissue generally accumulates higher levels of chromium compared to soft tissues. While liver and kidney are routinely analysed, they may not always serve as reliable indicators of Cr exposure. Therefore, the inclusion of bone, alongside liver and kidney, is recommended as a minimum requirement for future biomonitoring studies. Furthermore, Cr does not biomagnify in marine food webs involving mammals and other vertebrates (Outridge and Scheuhammer, 1993), suggesting that elevated Cr levels are more likely related to local exposure sources than trophic transfer.
Regression analysis of Cr content in tissues of all three dolphin species showed no statistically significant relationship between Cr concentration and variables such as body mass, age, sex, or location (p > 0.05). However, a significant association was found with the year of stranding. Chromium content shows a decreasing trend over time in all analysed tissues: muscle (regression coefficient b = 0.940), kidney (0.946), liver (0.928), spleen (0.945), and lungs (0.946). Additionally, in muscle tissue, a statistically significant negative correlation with body length was observed (b = 0.994, p = 0.017), indicating a decrease in Cr concentration with increasing dolphin body length (Table 3).
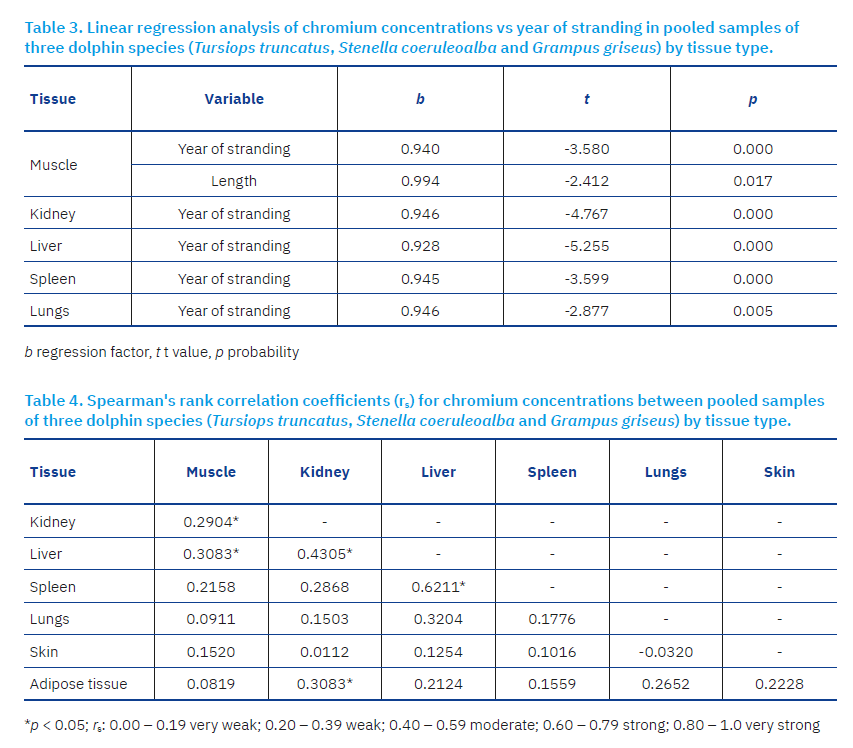
Spearman correlation analysis revealed several significant associations of Cr content among different tissues. A weak but statistically significant correlation was observed between muscle and kidney (rs = 0.2904*), and between muscle and liver (rs = 0.3083*). A moderate correlation was recorded between liver and kidney (rs = 0.4305*), while a strong correlation was found between liver and spleen (rs = 0.6211*). Additionally, a weak but statistically significant association was noted between blubber and kidney (rs = 0.3083*) (Table 4).
Data on Cr concentrations in cetaceans from the Mediterranean Sea remain limited, particularly for bottlenose, striped and Risso’s dolphins. In bottlenose dolphin, Cr concentrations measured in the kidney and lungs in this study were consistent with those previously reported for specimens from the Mediterranean (Bellante et al., 2009). In contrast, the hepatic concentration observed in this study (0.21 mg/kg) was approximately twice that reported for dolphins along the Italian coast (0.10 mg/kg), while the muscle concentration (0.25 mg/kg) was nearly half of the previously reported value (0.41 mg/kg). The elevated mean muscle concentration in the earlier study was influenced by a single individual from Sicily exhibiting an exceptionally high value (3.39 mg/kg) (Bellante et al., 2009).
Chromium concentrations in different tissues of striped dolphin, with the exception of adipose tissue, were comparable to those reported in individuals from southern Italy (Cardellichio et al., 2000), the Italian coastline during the period 2000–2009 (Bellante et al., 2009), and the Strait of Gibraltar (Rojo-Nieto and Fernandez-Maldonado, 2017). However, concentrations recorded in this study were up to sevenfold higher than those measured in dolphins from southern Italy in 1987 (Cardellichio et al., 2002). Mean concentrations in the kidney (0.21 mg/kg) and adipose tissue (0.50 mg/kg) were lower than those reported for the same species along the Italian coast (0.41 mg/kg and 0.88 mg/kg, respectively) (Bellante et al., 2009).
In Risso’s dolphin, Cr concentrations detected across all analysed tissues were considerably higher than values reported for specimens from the Italian coast (Bellante et al., 2009).
Chromium concentrations in the liver of striped dolphin in this study are comparable to other geographic regions worldwide, except for notably lower values observed in dolphins from the Brazilian and French Atlantic coasts (Holsbeek et al., 1998; Kunito et al., 2004). Data for other tissues are limited, and there are no records of Cr concentrations in lungs of striped dolphin. Kidney Cr levels in dolphins from the Iberian Peninsula (0.4 mg/kg) exceed those found here (0.21 mg/kg) (Mendez-Fernandez et al., 2014), while values in kidneys (0.05 mg/kg) and muscle (<0.0375 mg/kg) from the French Atlantic coast were significantly lower (kidney 0.21 mg/kg; muscle 0.21 mg/kg) compared to this study (Holsbeek et al., 1998). Muscle and adipose tissue Cr concentrations from dolphins off the coast of western Wales showed no major differences; however, an unusually high concentration was found in the melon (2.3 mg/kg) of a single individual (Morris et al., 1989).
Cr levels in the liver of Risso’s dolphin (0.35 mg/kg) correspond with the only available data from a specimen stranded on the English coast (0.26 mg/kg) (Law et al., 2001).
Elevated Cr concentrations in liver and kidneys of bottlenose dolphin have been reported worldwide, including the northwestern Iberian Peninsula (kidney 1.26 mg/kg; Mendez-Fernandez et al., 2014), South China Sea (liver 0.785 mg/kg; Parsons and Chan, 2001), Australia (liver 1.085 mg/kg; Law et al., 2003), and Hawaiian Islands (liver 1.53 mg/kg; Hansen et al., 2016). Most values align with this study except for notably lower liver (0.0625 mg/kg) and muscle (0.059 mg/kg) concentrations found in dolphins from the French Atlantic coast (Holsbeek et al., 1998).
In another coastal species, Pontoporia blainvillei, Cr concentrations reported by Kunito et al. (2004) from the Brazilian coast were substantially higher than those found by Panebianco et al. (2012) at the same location. Elevated Cr may be linked to temporary anthropogenic pollution from textile and leather industries; however, Cr in sediments was bound in unavailable chemical forms, limiting bioavailability (Panebianco et al., 2012).
Stavros et al. (2007) found no significant differences in Cr concentrations in the skin of bottlenose dolphin from two sites, one highly contaminated, suggesting effective homeostatic regulation or Cr being in an unavailable form. Similarly, Yang et al. (2002) reported that Cr in the skin of Phocoenoides dalli does not occur in the hexavalent form, which would otherwise cause dermatitis. These findings emphasise the importance of speciation analysis to assess chromium’s toxic effects in marine mammals (Stavros et al., 2007).
This study is the first investigation of Cr accumulation in the tissues of dolphins inhabiting the Croatian waters of the Adriatic Sea, aiming to fill existing gaps in understanding metal contamination in marine mammals. The analysis revealed significant interspecific differences in Cr concentrations in the kidney, liver, and spleen among the three dolphin species, suggesting that diet is a key factor influencing Cr accumulation. The results support previous findings on the impact of diet type on chromium levels in dolphin tissues; the highest Cr concentration was found in Risso’s dolphins, likely due to their consumption of cephalopods, which have higher Cr levels compared to fish.
No specific organ was identified as the most suitable for assessing Cr levels in this study. Given that Cr tends to accumulate in bone tissue, future research should include analysis of its concentrations in bones. Regression analysis showed no significant relationship between Cr levels and body mass, age, sex, or location, but chromium content significantly decreased over the years in all tissues examined. Additionally, Cr levels in muscle tissue declined with increasing dolphin body length.
In conclusion, the results obtained from three dolphin species allowed for robust statistical analysis, providing a more definitive assessment of Cr behaviour and regulation in the organs and tissues of these species. These findings contribute to a better understanding of the potential role of this trace element in marine mammals.
References [… show]
Analiza raspodjele kroma u tkivima dobrih, prugastih i glavatih dupina iz hrvatskog dijela Jadrana tijekom 20-godišnjeg razdoblja
Maja Đokić1, dokic@veinst.hr, orcid.org/0000-0002-3071-6208; Nina Bilandžić1, bilandzic@veinst.hr, orcid.org/0000-0002-0009-5367; Miroslav Benić2, benic@veinst.hr, orcid.org/0000-0001-7594-520X; Tomislav Bolanča3, tbolanca@fkit.unizg.hr, orcid.org/0000-0002-2268-8139; Tomislav Gomerčić4, tomislav.gomercic@vef.unizg.hr, orcid.org/0000-0001-7232-9428; Martina Đuras5, martina.duras@vef.hr, orcid.org/0000-0002-0538-0992; Marija Sedak1* (dopisni autor), sedak@veinst.hr, orcid.org/0000-0001-6861-0436.
1 Laboratorij za određivanje rezidua, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
2 Laboratorij za mastitise i kakvoću sirovog mlijeka, Odjel za bakteriologiju i parazitologiju, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
3 Zavod za analitičku kemiju, Fakultet kemijskog inženjerstva i tehnologije Sveučilišta u Zagrebu, 10000 Zagreb, Hrvatska
4 Zavod za veterinarsku biologiju, Veterinarski fakultet Sveučilišta u Zagrebu, 10000 Zagreb, Croatia
5 Zavod za anatomiju, histologiju i embriologiju, Veterinarski fakultet Sveučilišta u Zagrebu, 10000 Zagreb, Hrvatska
Sažetak
U ovom su istraživanju određene koncentracije kroma (Cr) u tkivima dobrih (Tursiops truncatus), plavobijelih (Stenella coeruleoalba) I glavatih dupina (Grampus griseus) — nasukanih uz hrvatsku obalu Jadranskog mora u razdoblju od 1995. do 2013. godine. Analizirano je ukupno 190 jedinki, uključujući 159 dobrih, 25 prugastih i 6 glavatih dupina. Koncentracije kroma određivane su u jetri, mišićima, bubrezima, koži, plućima, slezeni i masnom tkivu primjenom optičke emisijske spektrometrije s induktivno spregnutom plazmom (ICP-OES). Značajne međuvrsne razlike u koncentracijama kroma uočene su u bubrezima, jetri i slezeni, što upućuje na prehrambene navike kao glavni čimbenik nakupljanja kroma. Najviše koncentracije kroma utvrđene su kod glavatih dupina, što se može pripisati njihovoj prehrani glavonošcima, poznatima po višem sadržaju kroma u odnosu na ribu. Koncentracije kroma veće od 1 mg/kg zabilježene su u samo 16 uzoraka tkiva — od čega je 15 pripadalo dobrim dupinima, a jedan plavobijelom dupinu — što ukazuje na nisku ukupnu pojavnost povišenih razina kroma. Regresijskom analizom nije utvrđena značajna povezanost koncentracija kroma s tjelesnom masom, dobi, spolom ili geografskom lokacijom. Međutim, zabilježen je značajan pad koncentracija kroma u svim tkivima tijekom promatranog razdoblja.
Ključne riječi: krom, Tursiops truncatus, Stenella coeruleoalba, Grampus griseus, tkiva, Jadransko more

