Ergot alkaloids in cereal-based systems: Pathways of food and feed contamination, toxic effects and control measures
J. Pleadin*, I. Dželalija, D. Milićević, M. Zadravec, T. Murati, D. Petrović and I. Kmetič
Jelka Pleadin1* (corresponding author), pleadin@veinst.hr, orcid.org/0000-0002-0768-0462; Ivana Dželalija2, idzelalija5@gmail.com; Dragan Milićević2, dragan.milicevic@inmes.rs, orcid.org/0000-0001-9001-0584; Manuela Zadravec3, zadravec@veinst.hr, orcid.org/0000-0003-4382-4424; Teuta MURATI4 teuta.murati@pbf.unizg.hr, orcid.org/0000-0001-7157-400X; Danijela Petrović5, danijela.petrovic@aptf.sum.ba, orcid.org/0000-0003-4608-4767; Ivana KMETIČ4, ivana.kmetic@pbf.hr, orcid.org/0000-0001-5412-7128.
1Laboratory for Analytical Chemistry, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
2Institute of Meat Hygiene and Technology, 11000 Belgrade, Serbia
3Laboratory for Feed Microbiology, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
4Faculty of Food Technology and Biotechnology, University of Zagreb, 10000 Zagreb, Croatia
5Faculty of Agronomy and Food Technology, University of Mostar, 88000 Mostar, Bosnia and Herzegovina
https://doi.org/10.46419/cvj.57.4.7
Abstract
Ergot alkaloids (EAs) are a class of mycotoxins synthesised by various fungi, mainly by Claviceps purpurea and other members of the genus Claviceps. EAs are found in many economically important cereals, causing production losses in agriculture around the world. They are mycotoxins with well-documented toxicological effects on humans and animals, including vasoconstriction, neurotoxicity, hormonal disorders and impairment of reproductive function. Their presence in cereal crops, especially in rye, wheat and barley, poses a persistent risk to food and feed safety. Despite advances in analytical detection methods, the heterogeneous distribution of these alkaloids and the variability of their toxic profiles make effective risk assessment difficult. Chronic dietary exposure, even at subclinical levels, can lead to adverse health effects, emphasising the importance of continuous monitoring. Therefore, the establishment and enforcement of harmonised legal limits is essential, accompanied by comprehensive monitoring and the application of good agricultural and storage practises. A systematic and science-based control strategy is necessary to mitigate the risks associated with EAs and to ensure the protection of human and animal health within the food production chain. The aim of this review is to characterise EAs, their pathways of contamination, toxic effects in the body and control measures which should be applied for food and feed.
Keywords: mycotoxins, ergot alkaloids, cereals, mechanisms of action, toxicity, detection in food and feed
Introduction
Among the moulds of the genus Claviceps, the species Claviceps purpurea is the most widespread in Europe. C. purpurea parasitises the flowering heads of cereal plants (Tudzynski and Scheffer, 2004) and can produce ergot alkaloids (EAs) as mycotoxins with proven toxic effects on humans and animals, causing production losses in agriculture around the world (Pleadin et al., 2019; Singh et al., 2019; Carbonell-Rozas et al., 2023; Gurikar et al., 2023). EAs are present in many economically important cereals such as rye, wheat, triticale, barley, millet and oats, which are an important source of energy and an integral part of the diet of the human population, including infants, children, adolescents and the elderly, and must therefore comply with legal standards and be safe for health (Agriopoulou, 2021; Pleadin et al., 2022).
The toxicity of EAs is well known, as several epidemics of human poisoning have occurred from the consumption of contaminated grain throughout history. It is known that the contamination of cereals with sclerotia containing EAs can lead to significant economic losses due to crop failure, and the consumption of contaminated products can lead to ergot in humans and animals. Due to their proven toxicity, contamination of cereal grains with EAs remains a significant food and feed safety concern, necessitating regulatory monitoring and the development of analytical methods for their detection (EFSA, 2012). Monitoring should focus on the six predominant EAs (ergometrine, ergotamine, ergosine, ergocristine, ergocryptine and ergocornine, and their related –inines). EU Member States should, where possible, measure the content of sclerotia in the sample to improve knowledge of the relationship between the EAs contained in the sclerotia and in the whole grain (EFSA, 2017). Due to the stability of EAs and their resistance to degradation during processing, avoiding contamination at the field level is also crucial.
The aim of this review is to characterise EAs as mycotoxins that can occur in food and feed as a result of cereal contamination, to identify their main representatives, mechanisms of action and toxic effects in the body, and to provide an overview of the legislation and analytical methods for their detection in food and feed.
The main EAs and their properties
Ergot is a structurally diverse group of alkaloids derived from tryptophan and dimethylallyl pyrophosphate (DMAPP) (Jakubczyk et al., 2014). These mycotoxins represent a structurally diverse family of indole-based secondary metabolites with a common ergoline backbone and varying side chains. Their physicochemical properties, moderate basicity, and photosensitivity play a crucial role in the biological activity, toxicity and practical handling of this group of naturally occurring compounds derived from the ergoline ring system. Structurally, all EAs have a common ergoline core, a tetracyclic structure consisting of a fused indole ring, and a tricyclic system derived from lysergic acid. The basic structure, ergoline (C₁₆H₁₇N₂), serves as the backbone for a variety of substitutions that define each specific alkaloid (Flieger et al., 1997; Gerhards et al., 2014).
The physicochemical properties of EAs vary considerably depending on the substitution pattern. The molecular weights range from about 250 g/mol for simple clavins to over 600 g/mol for complex ergopeptins (Flieger et al., 1997). They have limited water solubility, but show higher solubility in organic solvents such as ethanol, methanol and chloroform (Krska and Crews, 2008). Most EAs are weak bases, typically with pKa values between 6.0 and 8.0, due to the presence of a tertiary amine. They are also moderately to highly lipophilic, with logP values between 1.5 and 4.0 depending on the compound (Flieger et al., 1997), affecting their absorption, distribution and bioavailability in biological systems.
EAs are chemically unstable when exposed to light, heat and alkaline pH and therefore require suitable storage conditions. They are more stable under acidic conditions, and the melting point is generally between 100 and 200°C. Due to the indole chromophore, EAs show strong UV absorption between 240 and 300 nm, which is useful for spectrophotometric detection. Pharmacologically, EAs exert their effect by interacting with serotonin (5-hydroxytryptamine; 5-HT), dopamine and adrenergic receptors, and act as partial agonists or antagonists (Flieger et al., 1997; Krska and Crews, 2008; Gerhards et al., 2014).
EAs are generally divided into three main structural classes. The first group, the clavin alkaloids, comprises relatively simple compounds with substituted ergoline rings and without peptide or amide side chains, such as agroclavine and elymoclavine. The second group consists of lysergic acid derivatives such as ergine (also known as LSA) and methylergometrine, which are characterised by simple amide side chains. The third and pharmacologically most active group are the ergopeptins, which contain a cyclic tripeptide moiety bound to lysergic acid. The best-known representatives of this group include ergotamine, ergocristine and ergovaline (Krska and Crews, 2008).
Mechanisms of action and effects in the body
The toxicity of EAs in humans and animals can lead to a condition known as ergotism, a severe toxic syndrome (Flieger et al., 1997; Krska and Crews, 2008), historically referred to as “St. Anthony’s Fire”, which manifests in two main forms: convulsive and gangrenous. The first person to identify ergot as the source of ergotism was the German physician Wendelin Thelius, who in 1596 described an epidemic that broke out in the Duchy of Hesse. In 1676, it was confirmed that the disease was caused by rye infected with C. purpurea. In 1778, the French physician Tessier ordered mandatory field drainage and crop cleansing after an epidemic that had broken out in Sologne, France that caused the death of over 8,000 people. Symptoms in general are characterised by hallucinations, muscle spasms, seizures and, in severe cases, loss of limbs due to vasoconstriction and tissue necrosis (Bové, 1970).
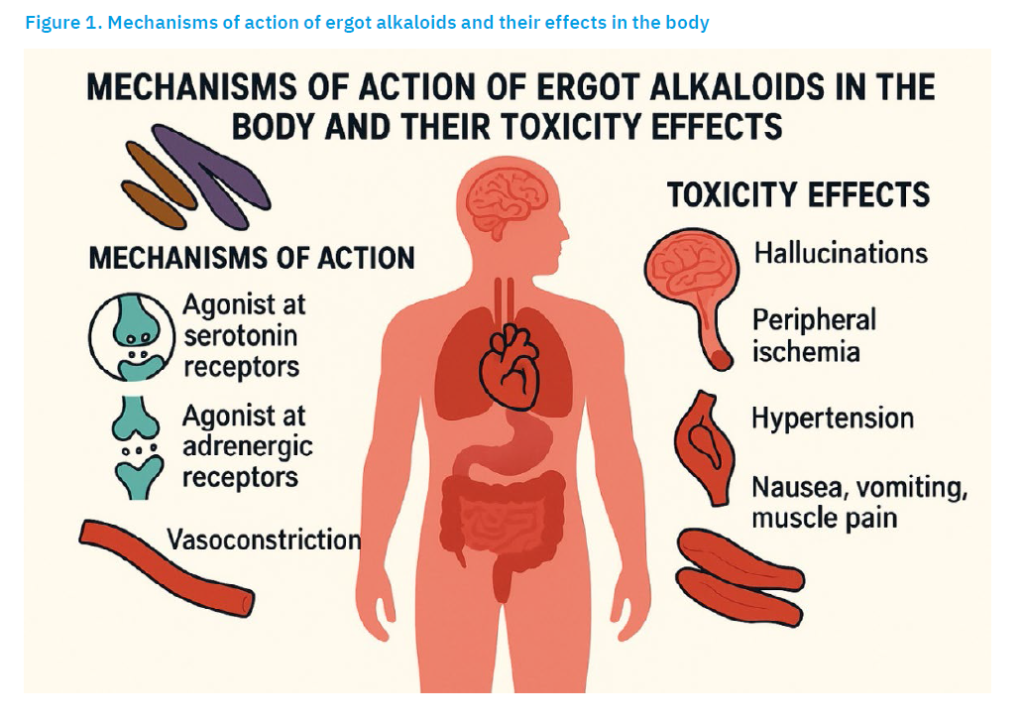
EAs are ligands that bind to specific receptors, in particular adrenergic, serotonergic and dopaminergic receptors (EFSA, 2012), thus influencing the role that the hormones adrenaline, serotonin and dopamine play in the body. However, due to the structural differences between EAs and adrenaline, serotonin and dopamine, EAs generally exhibit low specificity, i.e., selectivity in relation to the neuroreceptors mentioned and, depending on their individual structure, can exhibit complex behaviour as receptor agonists, partial agonists or antagonists. The great heterogeneity of adrenergic, serotonergic and dopaminergic receptors and the distribution of the various receptor types and subtypes in different tissues leads to a complex combination of biological reactions that correspond to the specific profile of each individual EA (EFSA, 2012). EAs have strong pharmacological effects on the nervous and vascular systems due to their structural similarity to neurotransmitters, such as dopamine, serotonin and noradrenaline (Flieger et al., 1997). Despite their toxicity, some EAs have pharmaceutical applications, i.e., ergotamine is used to treat migraines, while ergometrine is used to control postpartum haemorrhage due to its uterotonic effects (Krska and Crews, 2008). The most important underlying physiological mechanisms of the EAs and their effects in the body are shown in Figure 1.
The characteristic modes of their action include:
- Alpha-adrenergic receptor agonism: These compounds can activate alpha-adrenergic receptors, resulting in vasoconstriction. This property is utilised in certain medical conditions that require reduced blood flow (Willems et al., 2003).
- Interaction with serotonin receptors: EAs can also bind to various serotonin receptors (5-HT receptors), in particular to the 5-HT1B and 5-HT1D subtypes. This interaction can lead to vasoconstriction and modulation of neurotransmitter release, which is particularly relevant in the context of migraine treatment (Rushendran and Vellapandian, 2025).
- Vasoconstriction: The combined effect on serotonin and adrenergic receptors leads to vasoconstriction, which is the basis for its use in the treatment of migraine headaches (Goadsby, 2009). The vasoconstrictive properties help to counteract the vasodilation that occurs during a migraine attack.
- Dopamine receptor agonism: Many EAs, such as bromocriptine, act as agonists at dopamine receptors, particularly at subtype D2. This action can lead to effects on neurotransmission that are beneficial in the treatment of diseases such as Parkinson’s disease and hyperprolactinaemia (Elsworth, 2020).
- Inhibition of neuropeptide release: EAs can inhibit the release of neuropeptides such as CGRP (Calcitonin Gene-Related Peptide), which are involved in the pathophysiology of migraine. By modulating this pathway, EAs can alleviate migraine symptoms (Edvinsson et al., 2018).
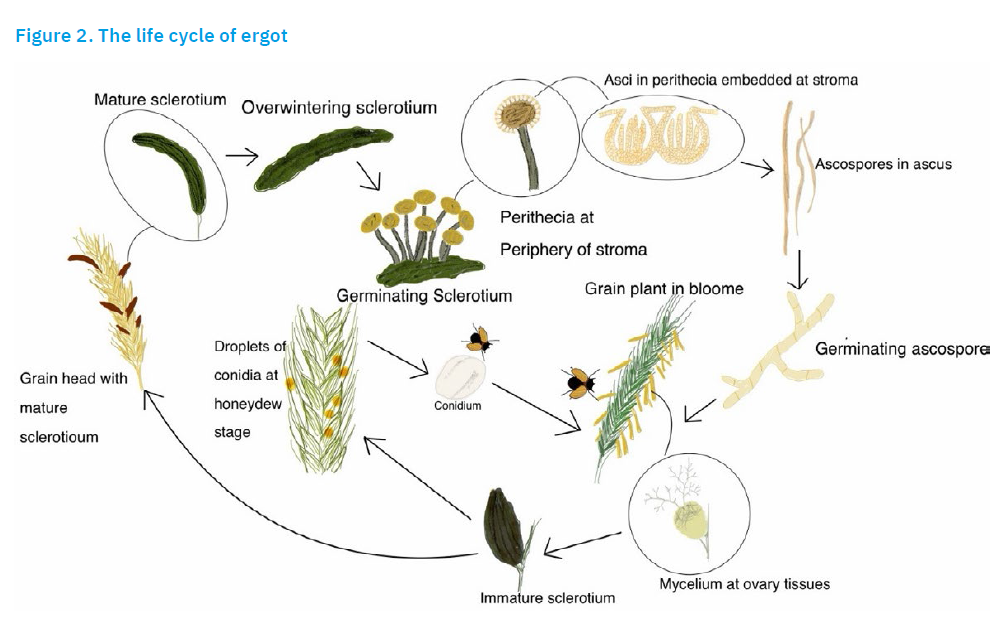
The ergot life cycle
Ergot is a disease of cereal and grass seed heads in which the grain is replaced by sclerotia (hardened masses of fungal mycelium) (Guo et al., 2016; Chung, 2021). C. purpurea produces two types of spores: sexual and asexual. Sexual spores, known as ascospores, are produced in stroma (compact masses of specialised vegetative hyphae) formed by the germination of sclerotia, which initiates the disease cycle. During germination, each sclerotium forms 1-60 stroma. Each stroma consists of a 0.5-2.5 cm high, flesh-coloured stalk with a spherical head at its tip. Numerous sexual fruiting bodies called perithecia develop at the periphery of each head, and each perithecium contains many sac-like cells called asci. Meiosis (cell division) produces eight long, multicellular ascospores in each ascus. On the other hand, asexual spores, the conidia, are produced after the ovary of the host plant has been infected with ascospores. Both types of spores are spread by wind, rain splashes or insects (Wegulo and Carlson, 2011). If their release coincides with the flowering period, the ascospores land on the flowers and infect the ovaries. One or more ovaries per head can be infected. A few days after infection, the ovary is replaced by a greyish-white mycelial mass (stroma) that occupies the space between the bracts of the flower. As the stroma grows and develops, the host plant produces a sticky, sweet secretion (honeydew) that covers the stroma. The conidia sprout from the stroma and float on the surface of the honeydew, attracting insects. The conidia then spread to other flowers and infect their ovaries. The honeydew phase lasts several days and is prolonged by damp and cloudy weather. The disease cycle is complete when the stroma, which has replaced each ovary at the head, matures into a sclerotia. The sclerotia fall to the soil before or during harvest or are introduced when contaminated grain is sown, starting the cycle again (Wegulo and Carlson, 2011). The ergot life cycle is presented in Figure 2.
Main producers and occurrence
During the harvest of cereals, seeds and grasses, the presence of sclerotia can lead to contamination of cereal-based food and feed. EA contamination can also occur at different stages of the food chain, as sclerotia can be broken up during transport, facilitating the entry of these compounds into the food chain (Chung, 2021; Sun et al., 2022). It has been demonstrated that a significant amount (up to 80%) (Lenain et al., 2012) of EAs in the food industry can be effectively removed by cleaning and size reduction techniques such as sizing, sorting and sieving. However, they cannot be eliminated completely, even with fungicides, which is why analytical methods for their determination are of great importance (Storm et al., 2008; Silva et al., 2023).
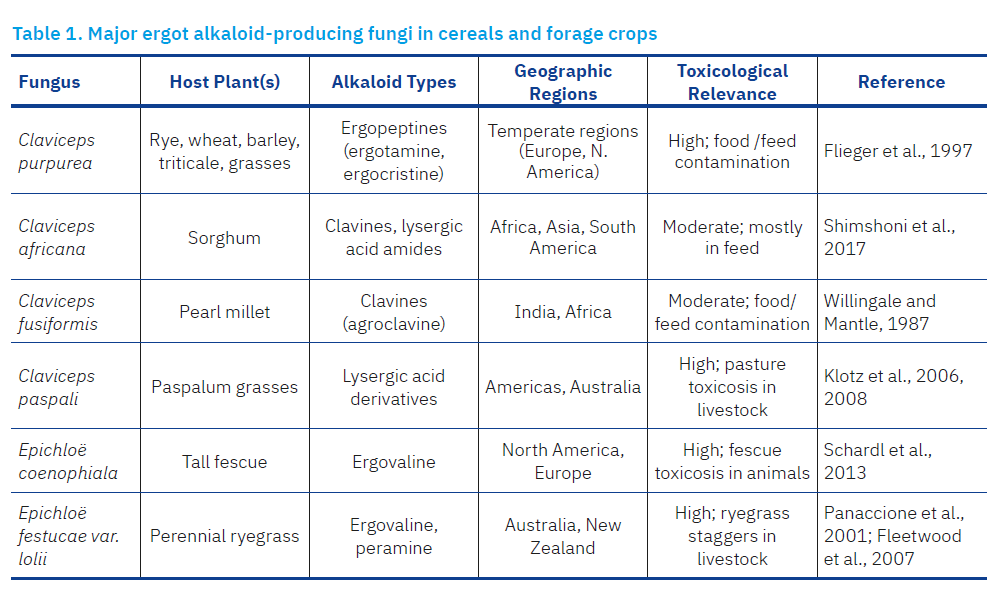
Major ergot alkaloid-producing fungi in cereals and forage crops are shown in Table 1. The genus Claviceps is the most widespread and economically important group of EA producers. It comprises over 50 described species, several of which are significant in agriculture and food safety. C. purpurea is the most widespread and economically important species, predominantly infecting rye (Secale cereale) but also wheat, triticale, barley, and other small-grain cereals (Flieger et al., 1997), forming toxic sclerotia in place of developing kernels. EAs in C. purpurea are found in sclerotia, with spectra varying between strains of this fungus (Lorenz et al., 2009). The main compounds produced include ergocristine, ergotamine, ergocornine, α- and β-ergocryptine, ergometrine, ergosine, ergocristinine, and ergosinine (Franzmann et al., 2010; EFSA, 2012). C. purpurea shows high morphological variability, as reflected in the different lengths and shapes of sclerotia, colour of the stomata, and the size and shape of conidia (Pažoutová, 2002).
Other important members of the genus Claviceps include species such as C. africana, in which dihydroergosine is the main EA present in its sclerotia; C. fusiformis, which is associated with the production of agroclavine, elimoclavine, hanoclavine, peniclavine and setoclavine, but not D-lysergic acid derivatives; and C. gigantea, C. paspali and C. hirtella, which are also known producers of clavines. In addition, strains of A. fumigatus and Penicillium, including P. roqueforti, P. verrucosum and P. commune, are also associated with the production of clavines. Aspergillus is known for the production of fumigaclavines A, B and C, while Penicillium is associated with the production of fumigaclavines A and B, but not C (Arroyo-Manzanares et al., 2017).
Claviceps africana is a pathogen of sorghum (Sorghum bicolor), mostly in tropical and subtropical regions. It produces clavine-type alkaloids and lysergic acid amides, though its toxicological significance is considered lower than that of C. purpurea (Shimshoni et al., 2017). C. fusiformis infects pearl millet (Pennisetum glaucum) and is commonly found in India and Africa. It produces agroclavine and elacomine, primarily clavine-type alkaloids (Willingale and Mantle, 1987). C. paspali is associated with ergot disease in Paspalum grasses (e.g., Paspalum dilatatum) and is particularly relevant in pastures. It synthesizes lysergic acid derivatives that can induce tremors and neurological disorders in livestock (Klotz et al., 2006, 2007, 2008). Some endophytic fungi of the genus Epichloë (previously Neotyphodium) form symbiotic associations with grasses and can also produce ergot alkaloids. Epichloë coenophiala infects tall fescue (Festuca arundinacea) and produces ergovaline, a potent ergopeptine alkaloid. Though beneficial for plant vigour and pest resistance, it can cause fescue toxicosis in grazing livestock (Schardl et al., 2013). In strains of the symbiotic endophyte Epichloë festucae, including var. lolii (Neotyphodium lolii) from ryegrass, 12 homologues have been found in a cluster that determines biosynthesis of ergovaline (Panaccione et al., 2001; Fleetwood et al., 2007).
EA production varies by geographic region, and C. purpurea is the primary producer in Europe (Gürbüzel et al., 2015; Malir et al., 2023). The synthesis of these compounds is influenced by several factors, including the type of fungus and plant, fungal concentration, temperature, humidity, and nutrient availability. Climatic conditions are of great importance, as EA synthesis is more favourable in moist soil and rainfall conditions (Guo et al., 2016; Carbonell-Rozas et al., 2021; de Sá et al., 2021). Cool, wet weather during flowering promotes infection by C. purpurea, while C. africana thrives in warm, humid climates. Some species are host-specific (e.g., C. fusiformis for pearl millet), while others have a broad host range, such as C. purpurea, which infects more than 400 grass species. Regulations in the EU limit the amount of sclerotia and total EAs in food and feed (EFSA, 2012). Additionally, infections in forage grasses by endophytic fungi can lead to chronic livestock disorders, reducing weight gain, fertility, and milk production (Klotz et al., 2007, 2008).
Toxicological studies
The toxic effects of EAs have been extensively studied. Only limited data are available for in vitro experiments on the toxic effects of individual EAs on human cell cultures (Mulac and Humpf, 2011). Most of the available data refer to receptor interaction experiments performed on dopamine-overexpressing cells or tumour cells (Larson et al., 1995, 1999). Further experiments showed a different toxic potential of peptide EAs compared to lysergic acid amide alkaloids, suggesting that the cytotoxicity of EAs in human cell lines is dependent on the specific type of alkaloid (Oda et al., 2008).
In vivo acute toxicity studies have shown different LD50 values (lethal dose, 50%) depending on factors such as the animal species, route of administration, and the specific EA (Arroyo-Manzanares et al., 2017). Griffith et al. (1978) reported a range of LD50 values (from 0.9 to 275 mg/kg body weight) for various natural and semi-synthetic EAs administered subcutaneously or orally to mice, rats and rabbits. Furthermore, the results show that rabbits are most sensitive, with LD50 values between 0.9 and 3.2 mg/kg body weight.
After oral intake of small amounts of EAs, acute symptoms such as vomiting, cramps, headaches and cardiovascular problems such as high blood pressure or cardiac arrhythmia can occur in humans. Functional disorders of the central nervous system can also occur. Even small intakes have been shown to trigger uterine contractions, which can lead to uterine haemorrhage and abortions. When large quantities are ingested, acute toxic effects can manifest in the form of circulatory disorders due to vasoconstriction of the blood vessels, which particularly affect the heart muscle, kidneys and extremities. These symptoms can be accompanied by hallucinations, convulsions, sensory disturbances and paralysis, and in severe cases can lead to respiratory or cardiac arrest (BfR, 2010).
There are two main forms of ergot, gangrenous and spasmodic. The gangrenous form is caused by the highly vasoconstrictive properties of some EAs, which lead to a restriction of blood flow in parts of the body (ischaemia). In the convulsive form, the tingling is accompanied by neurotoxic symptoms such as hallucinations, delirium and epileptic-like seizures. It has been hypothesised that vitamin A deficiency in combination with high concentrations of EAs could be a causative factor for convulsive ergotism. Other symptoms of ergotism include lethargy and depression (EFSA, 2012; Arroyo-Manzanares et al., 2017).
All animals are susceptible to ergot, but cattle are often the most severely affected. In cattle, ergot mainly affects the limbs, often resulting in the loss of ear and tail tips. Depending on the amount ingested, the feet and legs can also be affected, leading to lameness with possible swelling of the hocks and, in severe cases, loss of hooves. The altered circulation can affect thermoregulation and lead to heat intolerance. Affected cattle may exhibit symptoms such as rough coats and weight loss and may stand in water for extended periods of time or seek shade if available (Gupta, 2018). As EAs act on several neurotransmitter receptors, in particular adrenergic, dopaminergic and serotonergic receptors, EFSA (2012) considered neurotoxicity to be the main acute effect with symptoms such as restlessness, miosis or mydriasis (contraction and dilation of pupils), muscle weakness, tremors and stiffness in mammals. Long-term ingestion of moderate amounts of EAs can have a negative effect on reproduction, possibly causing abortions, lower birth weight and interruption of lactation.
Chronic oral ingestion of large amounts of EAs can lead to symptoms similar to those observed with the acute ingestion of large amounts. These observations are based on the adverse effects observed when certain EAs were used as active ingredients in pharmaceuticals, or when individuals became ill after consuming cereal products contaminated with EAs (BfR, 2010). However, there are currently no data on the chronic toxicity of EAs caused by human exposure to contaminated food. Only limited data are available on the use of ergot-containing medicinal products. In case studies on the long-term use of ergot medication for migraine headaches, severe claudication (limb pain) in the lower extremities due to chronic arterial insufficiency was reported (Garcia et al., 2000; Silberstein and McCrory, 2003; Fröhlich et al., 2010; Bogun et al., 2011). In all cases, treatment was discontinued and patients were asked to abstain from caffeine and cigarettes. Symptoms were successfully reversed with antiplatelet therapy. Korn et al. (2014) reported spontaneous alopecia, erosions, scabs and necrosis, especially in the tail region in rabbits, exclusively in young rabbits aged 113 ± 20 days fed hay and commercial pelleted feed. The results of the study suggest that EAs may have been the cause of tail necrosis. Immunoassays on blood samples revealed mean and maximum EA concentrations of 410 μg/kg and 1,700 μg/kg, respectively. In addition, EAs concentrations of up to 200 μg/kg were detected in the faeces of infected rabbits. The average and maximum dietary intake of total EAs was 17 and 71 μg/kg body weight, respectively. Other toxins were also detected in animal feed, but in quantities that, in the authors’ opinion, did not explain the observed effects.
Repeated administration of various EAs resulted in ischaemia, particularly in the extremities (e.g., tails) of rats, reduced body weight gain and changes in some hormone levels. Tail gangrene was observed in rats 5-7 days after a single intraperitoneal exposure to ergotoxin (a mixture of ergocornine, α- and β-ergocryptine and ergocristine) at a dose of 25 mg/kg body weight (Griffith et al., 1978). Repeated dose studies in rats showed no significant quantitative difference in the toxicity of ergotamine, ergometrine and α-ergocryptine, with NOAELs (no observed adverse effect levels) in the range of 0.22–0.60 mg/kg body weight per day (EFSA, 2012).
Regulatory limits in the EU
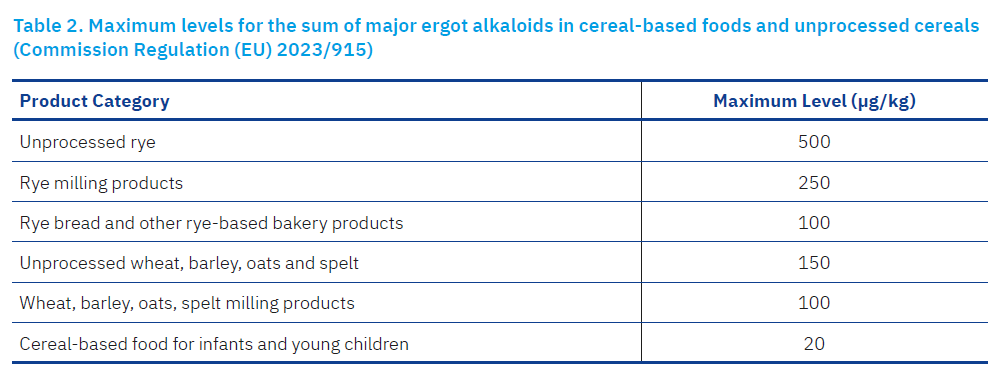
The European Union has set maximum levels for EAs in food and feed. These limits aim to reduce dietary exposure, especially for vulnerable groups such as children and pregnant women. The latest Commission Regulation (EU) 2021/1399 sets maximum levels for the sum of the 12 major ergot alkaloids (ergometrine, ergosine, ergotamine, ergocornine, ergocryptine, ergocristine and their corresponding epimers) in cereal-based foods and unprocessed cereals. The maximum EA level in foods (Table 2) is the lower limit of the sum of ergocornin/ergocorninin; ergocristin/ergocristinin; ergocryptin/ergocryptinin (form α and β); ergometrine/ergometrinin; ergosine/ergosinin; ergotamine/ergotaminin. These limits are based on a comprehensive risk assessment by the European Food Safety Authority (EFSA), which concluded that even low levels of chronic exposure could have adverse effects on human health, particularly in sensitive subgroups.
The EFSA Panel established an acute group reference dose of 1 μg/kg body weight and an acceptable daily intake for groups of total EAs of 0.6 μg/kg body weight (EFSA, 2012). They concluded that although the available scientific literature on EAs does not raise concerns in any population subgroup, the possible unknown effect of other food sources cannot be excluded (Commission Regulation, 2021). The Regulation stipulates that sampling and analysis protocols must follow harmonised standards to ensure uniform enforcement across Member States.
Commission Regulation (EU) 2023/915 on maximum levels for certain contaminants in foodstuffs entered into force on 25 April 2023 and repealed Regulation (EC) No 1881/2006. In addition, Directive 2002/32/EC of the European Parliament and of the Council on undesirable substances in animal feed, including EAs, has been in force since May 2002. Annex I of Commission Regulation (EU) 2023/915 sets maximum levels for mycotoxins in food, including EAs (expressed in μg/kg). The term ergot refers to the fungal structures of the species C. purpurea. Directive 2002/32/EC of the European Parliament and of the Council deals with undesirable substances in products intended for animal nutrition. According to this Directive, the maximum authorised level of alkaloids relating to cereals with a moisture content of 12% is 1,000 mg/kg (ppm). This applies to all feed containing unground cereals.
Detection and quantification methods
The identification of EAs is of great importance due to their presence in cereals and seeds and the potential health risks that their ingestion can cause. However, due to the large number of different foods and feeds, the diversity of compounds and their concentrations, it is a challenge to accurately determine EA concentrations in each type of sample (Carbonell-Rozas et al., 2022). This results in the need for an efficient and sensitive analytical method that allows the determination and quantification of these compounds below regulatory limits (Cigić and Prosen, 2009; Silva et al., 2023). The determination process usually involves several steps, starting from analyte extraction and sample clean-up to the detection and quantification of EAs and their epimeric forms (Crews, 2015).
The first step in identifying EAs in cereals is visual inspection of the whole grains for the presence of sclerotia, as sclerotia can be readily detected both with the naked eye and under the microscope (IAG, 2008; Pleadin et al., 2022). There are various methods for the qualitative and quantitative determination of EAs, including liquid chromatography (LC), enzyme-linked immunosorbent assay (ELISA), capillary electrophoresis (CE), gas chromatography (GC) and thin-layer chromatography (TLC) (Scott, 2007; Guo et al., 2016; Carbonell-Rozas et al., 2021). Liquid chromatography can be coupled with various detectors, including ultraviolet (UV) radiation, fluorescence detectors, evaporative light scattering detectors and mass spectrometry, while gas chromatography is usually coupled with electron capture detection (Guo et al., 2016; Veršilovskis et al., 2020; de Sá et al., 2021; Silva et al., 2023).
Chromatographic methods make it possible to determine the most important EAs individually and to calculate their sum to obtain the total EA content. They are based on the separation of components depending on their affinity to the mobile or stationary phase. Given their affinities, the components move differently in the column, resulting in their separation (Ülger et al., 2020). Liquid chromatographic methods such as thin-layer chromatography (TLC), high-performance liquid chromatography (HPLC) and ultra-high-performance liquid chromatography (UHPLC) are generally used to determine EAs. UHPLC has shown fast and efficient separation of compounds thanks to technological advances, using columns packed with submicron particles, justifying its application in the determination of mycotoxins (EFSA, 2012; Veršilovskis et al., 2020). As for detectors, UV light is used to quantify EAs, but it can trigger an epimerisation process that interferes with quantification. Therefore, fluorescent detectors have been used, not only because of the greater specificity and sensitivity, but also because some EAs are fluorescent by nature (Silva et al., 2023). Instead of analysing each EA individually, there is an alternative approach in which the EAs are converted into a common structure prior to analysis. This procedure is carried out by hydrolysis, in which the EAs and their epimers are split into a uniform structure – lysergic acid. This simplifies the analysis process as all compounds are processed as a single species, which can lead to savings in both time and resources (Höfs et al., 2023; Silva et al., 2023).
In view of the fact that EAs are not volatile and can decompose in the injector under the influence of high temperatures, gas chromatography is being used less frequently for the determination of these compounds. On the other hand, liquid chromatography is frequently used for analysing polar, non-volatile and thermolabile mycotoxins such as EAs (Müller et al., 2009; Lenain et al., 2012). In recent years, mass spectrometry in combination with chromatographic separation methods has become a standard method for the determination and quantification of EAs (Crews, 2015; Silva et al., 2023). Confirmation methods are crucial in laboratories for the accurate determination of EA concentrations.
However, there is a need for simpler and faster methods that can be applied at a lower cost and by less qualified analysts. One of the most important is the ELISA (enzyme-linked immunosorbent assay) method, which is based on the immunoenzymatic principle (Silva et al., 2023). ELISA is a sensitive and specific analytical method based on antigen-antibody interactions and is widely used for the detection and quantification of various contaminants, including mycotoxins, in food and feed. It offers a relatively rapid, low-cost and high-throughput alternative to chromatographic methods and is therefore suitable for routine screening purposes to control the quality and ensure the safety of food (Turner et al., 2009; Butorac et al., 2013). The main advantage of the ELISA method is its simplicity and portability compared to stationary chromatographic systems. At the same time, there is a risk of false-positive results due to cross-reactions or false-negative results due to the inhibition of antibodies by matrix components (Bonwick and Smith, 2004). Therefore, ELISA serves as a “screening” method, as it cannot definitively confirm the presence of EAs and further confirmation of the analysis is required for definitive results (Chung, 2021; Silva et al., 2023).
Conclusion
EAs pose a significant risk to human and animal health due to their strong toxicological effects, including vasoconstriction, neurotoxicity and reproductive disorders. Their presence in cereals, especially rye, wheat and barley, poses a serious problem for food and feed safety. Chronic exposure, even at low levels, can lead to ergot and have long-term health consequences. Given the complexity of ergot contamination and the variability of alkaloid profiles, systematic monitoring and control are essential. This includes the implementation of well-established monitoring programmes, the enforcement of legal limits, and the promotion of good agricultural and storage practises. Ensuring effective risk management is critical to protecting public health and maintaining the integrity of the food chain.
References [… show]
Ergot alkaloidi u sustavima na bazi žitarica: Putovi kontaminacije hrane i stočne hrane, toksični učinci i mjere kontrole
Jelka Pleadin1* (dopisni autor), pleadin@veinst.hr, orcid.org/0000-0002-0768-0462; Ivana Dželalija2, idzelalija5@gmail.com; Dragan Milićević2, dragan.milicevic@inmes.rs, orcid.org/0000-0001-9001-0584; Manuela Zadravec3, zadravec@veinst.hr, orcid.org/0000-0003-4382-4424; Teuta Murati4 teuta.murati@pbf.unizg.hr, orcid.org/0000-0001-7157-400X; Danijela Petrović5, danijela.petrovic@aptf.sum.ba, orcid.org/0000-0003-4608-4767; Ivana KMETIČ4, ivana.kmetic@pbf.hr orcid.org/0000-0001-5412-7128.
1Laboratorij za analitičku kemiju, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
2Institut za higijenu i tehnologiju mesa, 11000 Beograd, Srbija
3Laboratorij za mikrobilogiju hrane, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
4 Prehrambeno-biotehnološki fakultet Sveučilišta u Zagrebu, 10000 Zagreb, Hrvatska
5 Agronomski i prehrambeno-tehnološki fakultet Sveučilišta u Mostaru, 88000 Mostar, Bosna i Hercegovina
Ergot alkaloidi (EA) su skupina mikotoksina koje sintetiziraju različite plijesni, uglavnom rod Claviceps sa glavnom vrstom Claviceps purpurea. EA se nalaze u mnogim ekonomski važnim žitaricama i uzrokuju gubitke u proizvodnji u poljoprivredi diljem svijeta. U pitanju su mikotoksini s dobro dokumentiranim toksikološkim učincima na ljude i životinje, uključujući vazokonstrikciju, neurotoksičnost, hormonalne poremećaje i oštećenje reproduktivne funkcije. Njihova prisutnost u žitaricama, posebno u raži, pšenici i ječmu, predstavlja stalni rizik za sigurnost hrane i hrane za životinje. Unatoč napretku u analitičkim metodama detekcije, heterogena raspodjela ovih alkaloida i varijabilnost njihovih toksičnih profila otežavaju učinkovitu procjenu rizika. Kronična izloženost putem prehrane, čak i na subkliničkim razinama, može dovesti do štetnih učinaka na zdravlje, što naglašava važnost kontinuiranog praćenja. Stoga je uspostavljanje i provedba usklađenih zakonskih odrednica ključna, uz sveobuhvatno praćenje i primjenu dobrih poljoprivrednih i skladišnih praksi. Sustavna i znanstveno utemeljena strategija kontrole potrebna je za ublažavanje rizika povezanih s EA i za osiguranje zaštite zdravlja ljudi i životinja unutar lanca proizvodnje hrane. Cilj ovog pregleda je karakterizirati EA, putove kontaminacije, toksične učinke u tijelu i mjere njihove kontrole koje se trebaju primjenjivati na hranu i hranu za životinje.
Ključne riječi: mikotoksini, ergot alkaloidi, žitarice, mehanizmi djelovanja i toksičnost, detekcija u hrani i hrani za životinje

