Evaluation of microbiological safety and mycotoxin contamination of household produced meat products originating from Croatian indigenous pig breeds
V.Jaki Tkalec, M. Zadravec, A. Vulić*, Ž. Cvetnić, I. Kos, I. Vnučec, T. Lešić, N. Vahčić and J. Pleadin
Vesna JAKI TKALEC1#, jaki.vzk@veinst.hr, orcid.org/0000-0001-5944-7227; Manuela ZADRAVEC 2#, zadravec@veinst.hr, orcid.org/0000-0003-4382-4424; Ana VULIĆ*3 (corresponding author), vulic@veinst.hr, orcid.org/0000-0002-9379-7236; Željko CVETNIĆ4, zcvetnic@hazu.hr, orcid.org/0009-0009-6743-398X; Ivica KOS4, ikos@agr.hr, orcid.org/0000-0002-2126-2566; Ivan VNUČEC5, ivnucec@agr.hr, orcid.org/0000-0002-5190-3045; Nada VAHČIĆ 6, nvahcic@pbf.hr, orcid.org/0000-0003-1937-4873; Tina LEŠIĆ3, lesic@veinst.hr, orcid.org/0000-0001-6773-9473; Jelka PLEADIN3, pleadin@veinst.hr, orcid.org/0000-0002-0768-0462.
1Laboratory for Microbiology and Analytical Chemistry, Veterinary Department Križevci, Croatian Veterinary Institute, 48260 Križevci, Croatia,
2Laboratory for Feed Microbiology, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
3Laboratory for Analytical Chemistry, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
4Croatian Academy of Science and Art, 10000 Zagreb, Croatia
5Department of Animal Science, Faculty of Agriculture, University of Zagreb, 10000 Zagreb, Croatia
6Faculty of Food Technology and Biotechnology, University of Zagreb, 10000 Zagreb, Croatia
#both authors equally contributed to the study.
https://doi.org/10.46419/cvj.57.3.3
Abstract
This study aimed to assess the microbiological safety and mycotoxin contamination of traditional meat products made from Croatian indigenous pig breeds, produced in family households under uncontrolled conditions. Different factors, including natural, seasonal, and uncontrolled production conditions, pose challenges in maintaining consistent product microbiological and mycotoxin contamination. No bacteria from the genera Salmonella or Clostridium, or the species Staphylococcus aureus, were detected in any of the investigated samples. However, Listeria monocytogenes was found in a cured sausage sample from Black Slavonian pigs, rendering it unsafe for consumption. Additionally, Listeria innocua was identified in a dried sausage sample from Turopolje pigs. Yeast and mould contamination levels ranged as follows: cured sausages: 10² – 1.4 × 10⁴ cfu/g; whole ham: 1.4 × 10² – 2.7 × 10⁴ cfu/g; bacon: 2.6 × 10² – 3.5 × 10³ cfu/g. The dominant mould genus colonising the dry meat products was Penicillium (30 isolates), followed by Aspergillus (20 isolates), with Cladosporium and Mucor species present in lower numbers. The most frequently isolated Penicillium species were P. brevicompactum, P. commune, and P. solitum (85.7%), while the most common Aspergillus species were A. proliferans and A. tubingensis (57.1%). Regarding product type, bacon and ham met the respective safety standards, but sausages were contaminated with L. monocytogenes and L. innocua, making them unsafe for consumption. All products were safe in terms of mycotoxin contamination.
Key words: traditional meat products; safety; indigenous pig breeds; bacteria; moulds; physicochemical properties
Introduction
Pork meat and its various derived traditional meat products (TMPs) represent a significant component of the global diet. However, the increasing demand for pork and the subsequent intensification of production have resulted in a decline of indigenous pig breeds, which are typically raised extensively and exhibit lower yields (Lukić et al., 2020). Recent literature underscores the growing interest in preserving indigenous pig breeds, recognising their value as a genetic reserve (Pugliese and Sirtori, 2012). In Croatia, three indigenous pig breeds are particularly noteworthy: the Turopolje Pig (Turopoljska svinja) (TP), Black Slavonian Pig (Crna slavonska svinja) (BSP) and the Banovina Pig (Banijska šara) (BS) (Anonymous, 2023). These pig breeds are characterised by late sexual maturity, medium size, and a fat-meat type. They are highly resilient to environmental conditions, require minimal feeding demands, and are well-suited for extensive farming. Compared to hybrid pig breeds, they tend to accumulate fat more intensively. These pig breeds are primarily used in the production of various traditional meat products, such as bacon, ham, and sausages, crafted in family households. These products are regarded as culinary delicacies and represent a valuable part of the tourism and gastronomy offering (Pleadin et al., 2024).
The safety and quality of TMPs are influenced by a range of factors that also affect fresh meat, including pig genotype, rearing and feeding methods, and pre- and post-slaughter conditions. Additional elements, such as meat and fat selection, seasoning, and hygienic and environmental factors (e.g., temperature, fermentation, smoking, drying, and ripening), further contribute to the final product’s characteristics (Cerjak et al., 2011). The traditional production of local meat products takes place under natural and uncontrolled seasonal conditions, making it difficult to ensure consistency of produced products. Cured meat TMPs have traditionally been considered microbiologically safe due to their unique physicochemical properties. Key factors include low water activity (aw), low pH in products like fermented sausages, and the bacteriostatic and inhibitory effects of salt, nitrates, and nitrites, as well as antimicrobial metabolites from their natural microbiota (Menéndez et al., 2018). However, while rare, meat products have occasionally been linked to foodborne outbreaks. Pathogens such as Staphylococcus aureus, Listeria monocytogenes, Salmonella spp., and Clostridium botulinum, particularly in fermented sausages and hams, have been associated with such incidents, as reported worldwide (Lyytikäinen et al., 2000; Sofos, 2008; El Bayami et al., 2020; Duma et al., 2024).
Bacterial contamination is associated with fresh meat and handling during the early stages of TMP production, while mould contamination is more common during the ripening period. Although bacteria pose minimal health risks in cured meats, mould can be of significant concern. Household dry-cured TMPs often develop wild-type moulds that affect quality and safety, posing potential health risks. However, surface moulds also enhance aroma, flavour, and texture, retain moisture, provide antioxidants, stabilise colour, prevent rancidity, and inhibit microbial growth and surface defects such as slime or stickiness (Sunesen and Stahnke, 2003; Martín et al., 2004, 2006; Alapont et al., 2013; Lešić et al., 2021). However, during the months-long ripening process, mould metabolites, including mycotoxins, can penetrate the interior of the product. Mycotoxins, produced by certain mould species when triggered by environmental factors such as temperature or humidity, are potentially hazardous for consumption (Asefa et al., 2011; Pleadin et al., 2017). Production conditions (e.g., ripening temperature and time, relative humidity) and intrinsic product properties (e.g., pH, aw, salt content) affect the growth of desirable surface moulds (Asefa et al., 2011; Mediani et al., 2022). However, these conditions also allow the growth of undesirable, competing moulds. Contamination with competing moulds depends on production practices, the hygiene of the production area and ripening environment, and raw material quality. Undesirable moulds negatively impact quality in terms of deviations of organoleptic properties and surface staining, and can result in food safety issues, including mycotoxin contamination (aflatoxin B1 (AFB1) and ochratoxin (OTA)), and the presence of antibiotics responsible for allergic reactions and antibiotic resistance (Sunesen and Stahnke, 2003; Magista et al., 2017).
This study aimed to assess the microbiological safety, and the presence of mycotoxin contamination in TMPs derived from Croatian indigenous pig breeds. Results of the physicochemical properties of TMPs were also determined and linked with observed parameters of microbiological and mycological quality. Notably, this study is the first evaluation of microbiological and toxicological safety for meat products from these indigenous breeds.
Materials and Methods
Meat product samples
In total, 18 bacon samples (7 TP, 5 BSP, 6 BŠ), 16 dry-cured hams (6 TP, 5 BSP, 5 BŠ), and 21 dry-fermented sausages (9 TP, 6 BSP, 6 BŠ) were collected at the end of the production process. Samples were collected in minimum quantities of 1 kg per sample from nine family households (three households for each of three species) located in central and eastern Croatia.
Bacteriological analysis
Valid ISO standard methods were used for the detection of bacteria of the genus Salmonella, Listeria, coagulase-positive Staphylococci, sulfite-reducing bacteria and counting moulds and yeasts (ISO 6579-1:2017, ISO 11290-1:2017, ISO 6888-1:2021, ISO 15213:2004, ISO 21527-2:2012, respectively). Confirmatory tests for Listeria identification include catalase production (positive), Gram staining (Gram-positive rods) and additional tests such as rhamnose fermentation, haemolysis on blood agar, and the CAMP test. The automated Vitek2 Compact system (bioMerieux, Marcy-l’Étoile, France) with the Vitek 2 ID GP identification card was used for the identification of Listeria species.
Isolation and initial morphology-based mould identification
Mould isolation and identification were performed according to Lešić et al. (2020, 2021). Briefly, immediately after delivery of TMP samples to the lab, mould colonies visible on TMP surfaces were sampled using swabs and transferred to dichloran 18% glycerol agar (DG18; Merck, Darmstadt, Germany). After a 7-day incubation in darkness at 25 ± 1°C, isolates were purified (if needed) and preserved. The isolates were subsequently inoculated with DG18, malt extract agar (MEA; BD Difco, Franklin Lakes, NY, USA), and Czapek yeast extract agar (CYA; BD Difco, Franklin Lakes, NY, USA) and incubated for a further 7 days at 25 ± 1°C in darkness. After incubation, initial culture identification was performed based on the macro- and micro-morphological characteristics (Pitt and Hocking, 2009; Samson et al., 2019). The relative frequency of mould species was calculated as the ratio of the number of samples in which the mould species was present divided by the total number of samples, and multiplied by 100.
Molecular mould identification
The obtained isolates were also identified using molecular methods. Genomic DNA was extracted using the NucleoSpin Microbial DNA (Macherey-Nagel, Düren, Germany) according to manufacturer’s instructions. The primer Bt2a and Bt2b pair was used for amplification and sequencing of a part of the beta-tubulin (BenA) gene, and the primers Cmd5 and Cmd6 for partial calmodulin (CaM) gene amplification and sequencing for determining Aspergillus and Penicillium species, while ITS 1 and ITS 4 primers were used for the ITS region of other isolated moulds (Samson et al., 2019). After purification using the ExoSAP-IT PCR clean-up reagent (Affymetrix, Santa Clara, CA, USA), amplicons were sent to a commercial sequencing facility (Macrogen, Amsterdam, the Netherlands). Traces were assembled using the Seqman program in the Lasergene Package (DNASTAR, v.16; Madison, WI, USA). Strain identification was performed using the Basic Local Alignment Search Tool (BLAST) searches of the NCBI GenBank nucleotide database. In addition, the sequences were compared to the local sequence database housed at the Westerdijk Fungal Biodiversity Institute that contains all available Penicillium and Aspergillus reference sequences (Houbraken et al., 2020). The frequency of mould species was calculated as the ratio of each genus species and total isolated genus species.
Determination of physicochemical properties
For physicochemical analysis, samples were ground by means of a laboratory mill Grindomix GM 200 (Retsch, Haan, Germany). Water content and water activity were analysed immediately, and the rest of the sample was stored in the freezer at -20°C until analysis. Water activity was measured using an internal method using a measuring device (HygroPalm AW with Hygro Clip AW probe; EminTech, Lund, Sweden). The sample container was filled with the grounded sample up to the mark and placed in the device until the appearance of the visual signal signalising the end of measurement. pH value was determined according to the standard ISO 2917:2000. Briefly, 5.0 g homogenised sample was weighed into an Erlenmeyer flask and 50 mL 0.1 M KCl was added. The contents of the flask were shaken for 15 min on a horizontal shaker. After shaking, the pH value of the sample was measured using a pH-meter (Seven Compact, Scwerzenbach, Switzerland). For the determination of sodium, approximately 0.2 g sample was submitted to microwave acidic digestion (Ethos easy, Milestone, Italy) supported with hydrogen peroxide (7 mL 60% nitric acid and 3 mL hydrogen peroxide). Digested samples were quantitative transferred to volumetric flask and diluted with ultrapure water. Sodium was analysed by mean of flame atomic absorption spectroscopy (200 Series A4 with SPS 4 Autosampler, Agilent Technologies, Santa Clara, USA) at ʎ=589 nm with specific HC coded lamp specific for sodium (Agilent Technologies, Santa Clara, USA).
Determination of mycotoxins
Ultrapure water was obtained from a Direct-Q 3 UV device (Merck, Darmstadt, Germany). High purity chemicals for mycotoxin analysis were obtained from Honeywell (Charlotte, NC, USA) and p.a. chemicals from Sigma-Aldrich (St. Louis, MO, USA). Ultrapure nitric acid was obtained from Merck (Darmstadt, Germany). For sodium determination, a standard solution of 1000 μg/mL in 5% nitric acid (Agilent Technologies, Denver, CO, USA) was used for preparation of standards for the calibration curve. AFB1 toxin (2µg/mL in acetonitrile), AFB2 toxin (3 µg/mL in acetonitrile), AFG1 (2 µg/mL in acetonitrile) and AFG2 (0.5 µg/mL in acetonitrile) were supplied by Sigma-Aldrich Chemie GmbH (Darmstadt, Germany), while the crude OTA standard was obtained from LGC Standards (Luckenwalde, Germany). Mycotoxins (AFB1, AFB2, AFG1, AFG2 and OTA) were analysed by liquid chromatography-tandem mass spectrometry (LC-MS/MS). The high-performance liquid chromatograph (1260 Infinity, Agilent Technologies, Santa Clara, CA, USA) was coupled with a triple quadrupole mass spectrometer (6410 QQQ, Agilent Technologies, Santa Clara, CA, USA). The separation of analytes was achieved on a 150 ×4.6 mm, 5 μm particle size C18 Gemini analytical column (Phenomenex, Torrance, CA, USA), coupled with a SecurityGuard™ Cartridges Gemini C18, 4×3.0 mm ID pre-column (Phenomenex, Torrance, CA, USA). Immunoaffinity columns (AFLAOCHRAPREP®, R-Biopharm Rhône Ltd., Glasgow, Scotland) were used for extraction and sample clean up. The performance and validation of the method was previously described in detail by Lešić et al. (2022). The obtained LOD (limit of detection) values were as follows: OTA 0.18 μg/kg, AFB1 0.03 μg/kg, AFB2 0.03 μg/kg, AFG1 0.04 μg/kg and AFG2 0.05 μg/kg.
Results and Discussion
A key factor ensuring food safety is the absence of pathogenic bacteria or toxins that could pose a risk to human health. In this study, no bacteria from the genera Salmonella or Clostridium, or the species Staphylococcus aureus, were detected in any samples (Table 1). Fermented sausages are generally free of sulphite-reducing clostridia and coagulase-positive staphylococci (Comi et al., 2005; Rantsiou and Cocolin, 2006; Francesca et al., 2012). However, some studies have reported the isolation of suspected S. aureus colonies from meat mixtures (Rebecchi et al., 1998; Metaxopoulos et al., 2001), and Salmonella has been detected in 1.8% of meat products intended for raw consumption in European Union countries (EFSA and ECDC, 2017; da Silva Mutza et al., 2018).
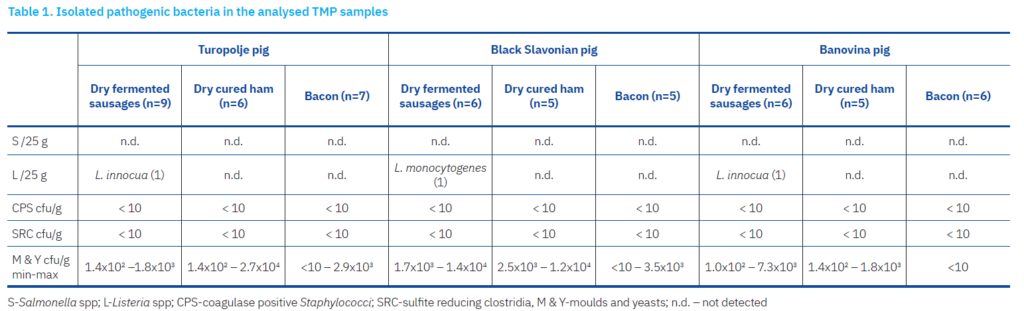
Among foodborne zoonoses, listeriosis has the highest mortality rate (EFSA, 2024). L. monocytogenes is the primary cause, most commonly affecting children, pregnant women, and immunocompromised individuals. L. ivanovii is also recognized as a pathogen, while L. innocua is considered potentially pathogenic (Orsi and Wiedmann, 2016; Gradovska et al., 2022; Bolten et al., 2024). In this study, Listeria monocytogenes was found in a sample of cured sausage from BSP, making the sample unsafe for consumption (EC 2073/2005). Listeria monocytogenes was not found in any of the other samples from the BSP, BS and TP breeds. L. innocua was identified in the dried sausage sample from TP (Table 1), but no Listeria was found in the samples from the other two breeds. L. monocytogenes, due to its wide distribution and resistance to various unfavourable growth and development conditions, is considered one of the most important foodborne pathogens (Kiš et al., 2019). It grows in a wide temperature range from 4 to 45°C, and although it does not form spores, it is resistant to various unfavourable growth conditions (low pH values and high NaCl concentrations) (Osek et al., 2022; Matle et al., 2024). It is assumed that products with a pH value ≤ 4.4 and an aw value ≤ 0.92 inhibit or prevent the growth of Listeria. In previous studies conducted in the Republic of Croatia, L. monocytogenes was isolated in 3.48% of samples of meat and meat products (Kiš et al., 2019), which is in line with other European countries (from 1.5% in Austria to 4.5% in Serbia; Kurpas et al., 2018). However, in this study, 11% sausages originated from TP, and 16% sausages originated from BŠ and BSP, respectively, were contaminated with Listeria spp.
Contamination of the analysed samples with yeasts and moulds (Table 2) ranged from 102 – 1.4×104 cfu/g in cured sausages, from 1.4×102 – 27×103 cfu/g in whole ham, and from 2.6×102 – 3.5×103 cfu/g in bacon. These results are in line with the results of Perković et al. (2022), who reported moulds and yeast contamination of BSP sausage in the range from 1.2×103 to 5.3×103. Moreover, Ambrosiadis et al. (2004) reported more than 1.0×105 cfu/g yeasts in Greek traditional sausages. These significant differences could be attributed to sample preparation, specifically whether the samples were used as a whole or with casing, bearing in mind surface overgrowth of yeast and mould. Yeasts are considered the main causative agent of spoilage of traditional sausages (Samelis and Metaxopoulus, 1998). Additionally, Plavšić et al. (2015) reported an average contamination of dried smoked products (type not specified) with mould contamination at 1.2×106 cfu/g and yeast at 1.0×107 cfu/g. These observations are contrary to the resulted presented here and the study of Karabiyikli et al. (2015), who reported contamination of yeasts and moulds up to 1.5×104 cfu/g. It is worth mentioning that there is currently no consensus or legislation in the European Union regarding the criteria for moulds in meat products, nor are they prescribed by the national guidelines in Croatia.
Previous studies (Zadravec et al., 2020, 2023) identified Penicillium as the dominant mould genus colonising the surfaces of Croatian dry-cured TMPs, while species of the genera Aspergillus, Cladosporium and Mucor are present in significantly lower numbers. In the current study (Table 2), Penicillium (30 isolates) was the dominant mould genus colonising the surfaces of studied dry meat products, followed by Aspergillus (20 isolates), while Cladosporium and Mucor species appear in significantly lower numbers (two and one isolate, respectively). The occurrence and diversity of these moulds are closely linked to TMP production technology, particularly the season and ripening period (Perković et al., 2022; Zadravec et al., 2023).
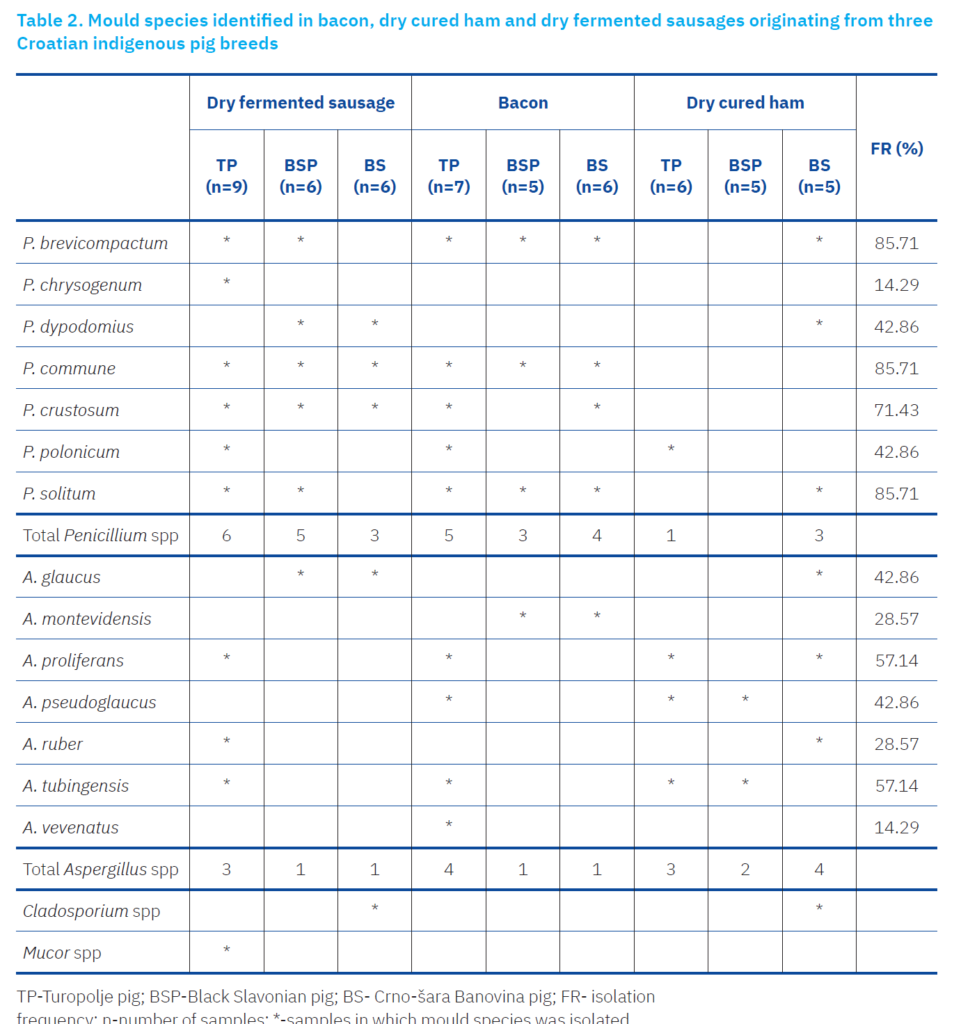
Penicillium species were predominant in all TMPs. Most isolates were obtained from dry fermented sausages and bacon, from which 12 Penicillium species were identified. The most frequently isolated species were P. brevicompactum, P. commune, and P. solitum (85.7%), followed by P. crustosum (71.4%), P. polonicum and P. dypodomius (42.9%), and P. chrysogenum (14.3%). The predominance of Penicillium aligns with previous research on TMPs, especially studies on dry-fermented sausages, as most Penicillium species are psychrotolerant and thrive in the winter ripening conditions characteristic of these products (Rodrigues et al., 2019; Vila et al., 2019, Perković et al., 2022). Additionally, most of the Penicillium species were isolated from sausages, most commonly in TP (six species), then in BSP (five species), and least in BŠ (three species). Bacon surfaces of TP were most overgrown with different mould species (five Penicillium and four Aspergillus species). Contrary to sausages and bacon, the surface of dry cured ham housed Aspergillus species, among them A. proliferans and A. tubingensis were isolated at a frequency of 57.1% respectively, followed by A. glaucus and A. pseudoglaucus (42.9%), A. montevidensis and A. ruber (28.6%) and A. vevenatus (14.3%). Zadravec et al. (2023) also reported A. proliferans as the most frequently isolated species from TMPs. Bacon and sausages contained 14 and 12 Penicillium species, respectively, while four Penicillium species and nine Aspergillus species were identified on the ham surface. These results could be explained by the fact that hams ripen during the warmer part of the year, which is more suitable for Aspergillus species. The same findings were observed by Comi et al., (2004), Zadravec et al., (2020) and Lešić et al., (2021), in which Aspergillus species were predominant over Pencillium species on prosciutto and ham surfaces.
Variations in the production process and a lack of standardisation can lead to the contamination of meat products with mycotoxins. The occurrence of these contaminants is influenced by the varying hygienic and environmental conditions that facilitate the growth of specific microbial flora and, consequently, the production of mycotoxins. Numerous studies have reported contamination of TMPs with mycotoxins (Pleadin et al., 2021), and the possible pathways of contamination. To date, there is no consensus or legislation in the European Union regarding maximum reference limits for mycotoxins in meat products; only a few countries have established national limits for ochratoxin A. TMPs produced in family households are more susceptible to microbiological contamination due to the less-controlled production process compared to industrial production. It should be noted that in the presented study, none of the AFs or OTA producers were isolated.
All samples from this study were analysed for the presence of AFs, as mycotoxins of the highest toxicity, and OTA, a mycotoxin that has been detected in a variety of meat products (Pleadin et al., 2015; Sánchez-Montero et al., 2019; Chen et al., 2022; Stefanello et al., 2022). Even though no mycotoxin-producing moulds were isolated, there is still a possibility that mycotoxins produced when moulds encounter unfavourable conditions could be present in dry-cured meat products. This may occur even if the original mould producers have died or been overgrown by other moulds (Alonso et al., 2013; Pleadin et al., 2016). Contamination can also result from spices, other raw materials, or carry-over effects from farm animals exposed to contaminated feed (Pleadin, 2022). None of the TMPs analysed in this study were contaminated with AFs or OTA, indicating that there was no contamination through any of the possible direct or indirect pathways.
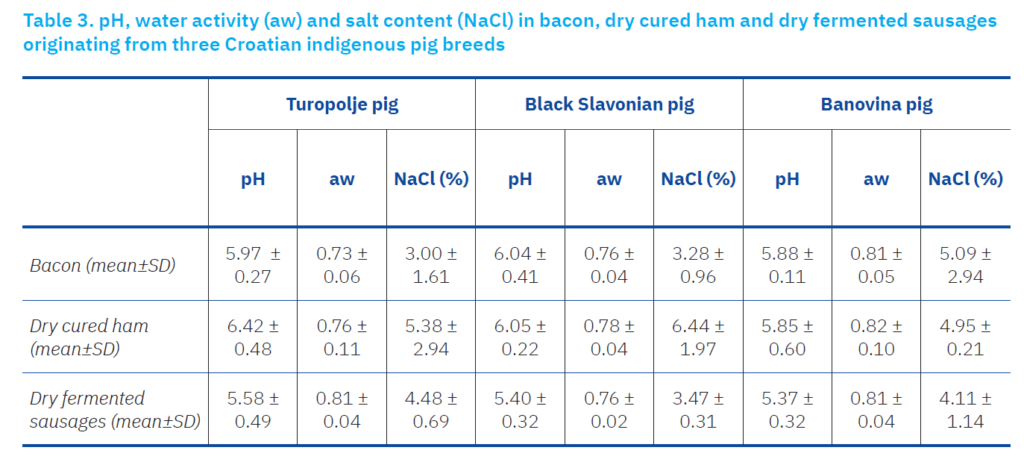
During the fermentation and ripening processes of meat products, proteolytic and lipolytic activities result in a decrease in pH value and aw (Lešić et al., 2020), which is important for product safety regarding microbiological contamination, and for the stability of products during storage. pH is an indicator of fermentation and ripening, primarily resulting from the activity of lactic acid bacteria. The pH values in bacon samples ranged from 5.88 to 6.04, in dry-cured hams from 5.85 to 6.42, and in dry-fermented sausages from 5.37 to 5.58 (Table 1), with no significant differences among the same meat products originating from different pig breeds. The values obtained for dry-fermented sausages align with the pH values for low-acid fermented meat products (final pH, 5.3 to 6.2) (Aymerich et al., 2003), but did not suppress the growth of Listeria reported here. Bacon samples displayed higher pH values compared to dry-fermented sausages, which can be attributed to the longer ripening period, consistent with findings from other studies (Guo et al., 2016).
Dry-cured hams undergo an extended ripening period, during which a significant decrease in pH is observed. Like other low-acid meat products, the expected final pH value at the end of the ripening process is below 6.2. In the current study, the group of dry-cured hams from TP exhibited a higher average pH value (6.42 ± 0.48) compared to other groups and previous studies (Table 3). This discrepancy may be attributed to a lack of standardisation in the production process.
The aw values for all meat samples in this study were below 0.85, indicating unfavourable conditions for the growth of microorganisms, with the exception of moulds from the Penicillium and Aspergillus genera, which favour lower aw values (Pitt and Hocking, 2009). The aw values ranged from 0.73 to 0.81 in bacon samples, from 0.76 to 0.82 in dry-cured hams, and from 0.76 to 0.81 in dry-fermented sausages (Table 3). These values are consistent with the production process concerning fermentation and ripening, during which a decrease in aw occurs, as reported elsewhere (Lešić et al., 2020, 2021). Similar to the pH values, no significant differences were observed between meat products from different pig breeds.
Another important factor influencing microbiological safety is the salt content, which creates unfavourable conditions for the growth of microorganisms. As expected, the lowest salt content was found in bacon samples, with the exception of bacon from BŠ, where the average salt content exceeded 5 g/100 g, as opposed to approximately 3 g/100 g in the other two groups. The salt content varied from 4.95 g/100 g to 6.44 g/100 g in dry-cured hams and from 4.11 g/100 g to 5.37 g/100 g in dry-fermented sausages. That fact can explain that most of the isolated Aspergillus species in the presented study belong to the section Aspergillus, which are more xerophilic species, hence in the presence of 5% salt (Chen et al., 2017).
Conclusions
The traditional local production of TMPs occurs under natural and uncontrolled seasonal conditions, making it challenging to ensure consistent quality and safety. However, traditional processing techniques and natural preservation methods can affect the safety of certain products. In the present study, dry-cured meat products such as bacon and ham, originating from three indigenous Croatian pig breeds, were found to be safe for consumers despite being produced under uncontrolled conditions. No contamination with pathogenic bacteria, moulds, yeasts, or mycotoxins was detected in any of the tested TMPs, highlighting the effectiveness of traditional curing and preservation practices. The only exception pursuant to the Regulation was a sample of dry-fermented sausage from the Black Slavonian pig breed, which was found to be unsafe for human consumption due to contamination with L. monocytogenes. This finding underscores the need for improvement in hygiene and production practice to mitigate the risk of contamination and ensure consumer safety.
Acknowledgment
This research was funded by the Croatian Academy of Sciences and Arts under the project with grant number 40-40/01-13/2023, and co-financed by Zagreb County and the Croatian Veterinary Institute, Zagreb, Croatia.
References [… show]
Procjena mikrobiološke sigurnosti i kontaminacije mikotoksinima u mesnim proizvodima proizvedenim u kućanstvima, koji potječu od autohtonih pasmina svinja iz Hrvatske
Vesna JAKI TKALEC1#, jaki.vzk@veinst.hr, orcid.org/0000-0001-5944-7227; Manuela ZADRAVEC2#, , zadravec@veinst.hr, orcid.org/0000-0003-4382-4424; Ana VULIĆ*3 (dopisni autor), vulic@veinst.hr, orcid.org/0000-0002-9379-7236; Željko CVETNIĆ4, zcvetnic@hazu.hr, orcid.org/0009-0009-6743-398X; Ivica KOS5, ikos@agr.hr, orcid.org/0000-0002-2126-2566; Ivan VNUČEC 5, ivnucec@agr.hr, orcid.org/0000-0002-5190-3045; Nada VHAČIĆ 6, nvahcic@pbf.hr, orcid.org/0000-0003-1937-4873; Tina LEŠIĆ3, lesic@veinst.hr, orcid.org/0000-0001-6773-9473; Jelka PLEADIN3, pleadin@veinst.hr, orcid.org/0000-0002-0768-0462.
1Laboratorij za mikrobiologiju i analitičku kemiju, Veterinarski zavod Križevci, Hrvatski veterinarski institut, 48260 Križevci, Hrvatska
2 Laboratorij za mikrobiologiju hrane za životinje, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Croatia
3Laboratorij za analitičku kemiju, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Croatia
4Hrvatska akademija znanosti i umjetnosti, 10000 Zagreb, Hrvatska
5Odsjek za animalne znanosti, Agronomski fakultet Sveučilišta u Zagrebu, 10000 Zagreb, Hrvatska
6Prehrambeno-biotehnološki fakultet Sveučilišta u Zagrebu, 10000 Zagreb, Croatia
#oba autora su podjednako doprinijela istraživanju.
Ovo istraživanje imalo za cilj je imalo procijeniti mikrobiološku sigurnost i kontaminaciju mikotoksinima tradicionalnih mesnih proizvoda izrađenih od hrvatskih autohtonih pasmina svinja, proizvedenih u obiteljskim domaćinstvima pod nekontroliranim uvjetima. Nedavna istraživanja ističu rastući interes za očuvanjem autohtonih pasmina svinja zbog njihove vrijednosti kao izvora genetske raznolikosti. Te pasmine uglavnom se koriste za proizvodnju tradicionalnih mesnih proizvoda poput slanine, pršuta i kobasica, a koje su obično izrađene u obiteljskim domaćinstvima. Međutim, prirodni, sezonski i nekontrolirani uvjeti proizvodnje predstavljaju izazove u održavanju dosljedne mikrobiološke sigurnosti i kontrole kontaminacije mikotoksinima. U ispitivanim uzorcima nisu otkriveni bakterije iz roda Salmonella ili Clostridium, niti Staphylococcus aureus. Međutim, Listeria monocytogenes je pronađena u uzorku suhe kobasica od crne slavonske svinje, čime je proizvod postao nesiguran za konzumaciju. U uzorku suhe kobasice od turopoljske svinje identificirana je isto tako Listeria innocua. Razine kontaminacije kvascima i plijesnima kretale su se u rasponu: suhe kobasice 10² – 1,4 × 10⁴ CFU/g; cijeli pršut 1,4 × 10² – 2,7 × 10⁴ CFU/g; slanina 2,6 × 10² – 3,5 × 10³ CFU/g. Najdominantniji rod plijesni koji je kolonizirao suhe mesne proizvode bio je Penicillium (30 izolata), zatim Aspergillus (20 izolata), dok su vrste Cladosporium i Mucor bile prisutne u manjim brojevima. Najčešće izolirane vrste Penicillium bile su P. brevicompactum, P. commune i P. solitum (85,7 %), dok su među vrstama Aspergillus najčešći A. proliferans i A. tubingensis (57,1 %). Bez obzira na pasminu, slanina i pršut održavali su jednaku razinu sigurnosti, no kobasice su bile kontaminirane L. monocytogenes i L. innocua, što ih čini nesigurnima za konzumaciju. Svi proizvodi bili su sigurni u pogledu kontaminacije mikotoksinima.
Ključne riječi: tradicionalni mesni proizvodi, sigurnost, autohtone pasmine svinja, bakterije, plijesni, fizikalno-kemijska svojstva

