First documented case of canine distemper virus in a leopard cub (Panthera pardus) in Tamil Nadu, India
N.V. Rajesh*, S. Manoharan, S. Muthukrishnan, S. Jaisree, P. Jayashree, E. Madhesh, R. B. Vishnurahav, M. Ranjithkumar, R. C. Sundararajan, E. Tamileniyan
Nakulan Valsala Rajesh1* (corresponding author), crocvet@gmail.com, orcid.org/0000-0003-0399-9051; Shunmugavel Manoharan2, smanovet@yahoo.com, orcid.org/0009-0005-7520-8281; Sundaram Muthukrishnan3, muthukrishnan.s@tanuvas.ac.in, orcid.org/0009-0004-2901-5342; Srinivasan Jaisree4, drjai81@yahoo.com, orcid.org/0000-0003-4962-620X; Panneerselvam Jayashree5, pjayashree2003@gmail.com, orcid.org/0009-0002-1888-8709; Elayaraman Madhesh1, madheshvmd@gmail.com, orcid.org/0000-0003-2137-9016; Radhakrishnan Bhuvaneshwari Vishnurahav6, vishnu.vet6@gmail.com, orcid.org/0000-0003-1962-4238, Muthusamy Ranjithkumar1, clmranjith@gmail.com, orcid.org/0000-0001-8389-4633; Rajagopalan Chandrasekaran Sundararajan1, rcsundararajan@gmail.com, 0000-0002-0843-1310; Elangovan Tamileniyan7 tamileniyan.2000@gmail.com, orcid.org/0009-0003-2459-9847.
1Department of Veterinary Medicine, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
2Forest Veterinary Assistant Surgeon, Forest Veterinary Unit, Forest Extension Division Campus, Ponnakudi, 627 151Tirunelveli, India.
3Department of Veterinary Anatomy, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
4Central University Laboratory, Centre for Animal Health Studies, Madhavaram Milk Colony, 600 051Chennai, India.
5UG Scholar, Veterinary College and Research Institute, 627 358Tirunelveli, India
6Department of Veterinary Clinical Complex, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
7Internee, Veterinary College and Research Institute, 614 625 Orathanadu, India.
https://doi.org/10.46419/cvj.57.4.9
Abstract
A four-month female leopard cub (Panthera pardus), rescued from a residential area in Kaliyal, Kanyakumari, Tamil Nadu, India with exhibited sign of dullness, convulsions, seizures, and profuse salivation was screened for infectious disease conditions and confirmed as canine distemper (CD) by conventional PCR method. To our best knowledge this was the first documented case of CD in a leopard in Tamil Nadu, India and this report focused on the integrative treatment with allopathic and siddha medicine for the disease condition.
Keywords: allopathic treatment; canine distemper; leopard; siddha treatment; Tamil Naduž
Introduction
India hosts a diverse range of wild felids, including the ecologically important leopard (Panthera pardus). Declining populations have led to their protection under Schedule I of the Wildlife Protection Act, 1972, and Appendix I of CITES (Kadam et al., 2022). The IUCN Red List (2014) classifies these species as Vulnerable due to habitat loss, human–wildlife conflict, and poaching. In addition to these threats, the emergence of infectious diseases such as canine distemper (CD) poses a severe challenge to their survival.
CD is a highly contagious viral disease caused by a canine morbillivirus – canine distemper virus (CDV), a single-stranded, negative-sense RNA virus of the genus Morbillivirus International Committee on Taxonomy of Viruses (ICTV). Morbillivirus. ICTV Virus Taxonomy, (2021). It primarily affects canids but is capable of infecting numerous mammalian families (Lan et al., 2005), including Felidae, Hyaenidae, Mustelidae, Ursidae, and even species within Artiodactyla, Primates, Proboscidea, and Rodentia (Debesa Belizário Granjeiro et al., 2020; Kličková et al., 2022). CDV has been increasingly reported in wild felids (Gilbert et al., 2014), raising significant conservation concerns.
In India, the first CDV outbreak in wild felids was recorded in Asiatic lions in Gujarat (Mourya et al., 2019), followed by molecular and pathological detections in several states (Kadam et al., 2022). To our knowledge, this paper presents the first documented case of CDV in a wild rescued wild leopard cub from Tamil Nadu, India.
Animals infected with CDV commonly exhibit respiratory, gastrointestinal, dermatologic, and neurological involvement (Lan et al., 2005). Transmission occurs through direct contact with infected body fluids or contaminated environments (Khorozyan et al., 2015). Recent CDV outbreaks in wild felids, including leopards and tigers in India, pose a growing conservation threat. Clinically, CD often presents with biphasic fever, serous to mucopurulent oculo-nasal discharge, anorexia, conjunctivitis, bronchitis, pneumonia, gastroenteritis, neurological signs, and skin lesions such as dermatitis or hyperkeratosis (Thulin et al., 1992; Kadam et al., 2022).
Given the susceptibility of wild felids and their vulnerable status, enhanced surveillance and disease management are crucial. As treatment for viral diseases is often unsuccessful, this report describes a rescued four-month-old wild leopard cub from southern India, outlining its clinical signs, diagnosis, integrated allopathic–Siddha treatment, and disease outcome.
Case presentation
A rescued four-month-old female leopard cub weighing 8.5 kg, from the Kaliyal region of Kanyakumari district (8°26′44.4948″ N, 77°14′58.0092″ E), southern Tamil Nadu, India, exhibited severe neurological and systemic signs, including dullness, convulsions, intermittent seizures, and profuse salivation (Fig. 1).

Clinical examination revealed open-mouth breathing (Fig. 2), hyperkeratosis of the foot pads (Fig. 3), muscle twitching, and serpentine tongue movements.


Blood and nasal, oral and rectal swab samples were collected from the live cub. Following death, tissue samples from the cornea, brain, heart, liver, lungs, and spleen were obtained during post-mortem examination. Blood, nasal and oral swabs, and tissue samples from the brain, lungs and spleen were submitted to the Central University Laboratory (CUL), Madhavaram, Tamil Nadu, for the diagnosis of canine distemper. For rabies diagnosis, corneal and brain tissues were subjected to fluorescent antibody testing (FAT) and RT-PCR at CUL. To rule out haemoprotozoan parasitic infections, blood smears and blood samples were analysed by PCR at the Translational Research Platform for Veterinary Biologicals (TRPVB), Madhavaram. Additionally, blood samples were processed for haematological and serum biochemical analyses following standard protocols (Coles, 1986) at CUL. Gut contents (25 g) and liver samples (25 g) were submitted to the Regional Forensic Science Laboratory, Tirunelveli, for toxicological analysis. Rectal swab and heart tissue samples were not submitted for CDV screening by PCR and were retained at the Department of Veterinary Medicine, Tirunelveli.An integrated treatment approach using allopathic and siddha medicine was attempted; however, the animal collapsed on the 19th day of treatment. This study discusses the treatment protocol and therapeutic outcome, laboratory findings of the collected samples, and the possible reasons for treatment failure.
Investigations
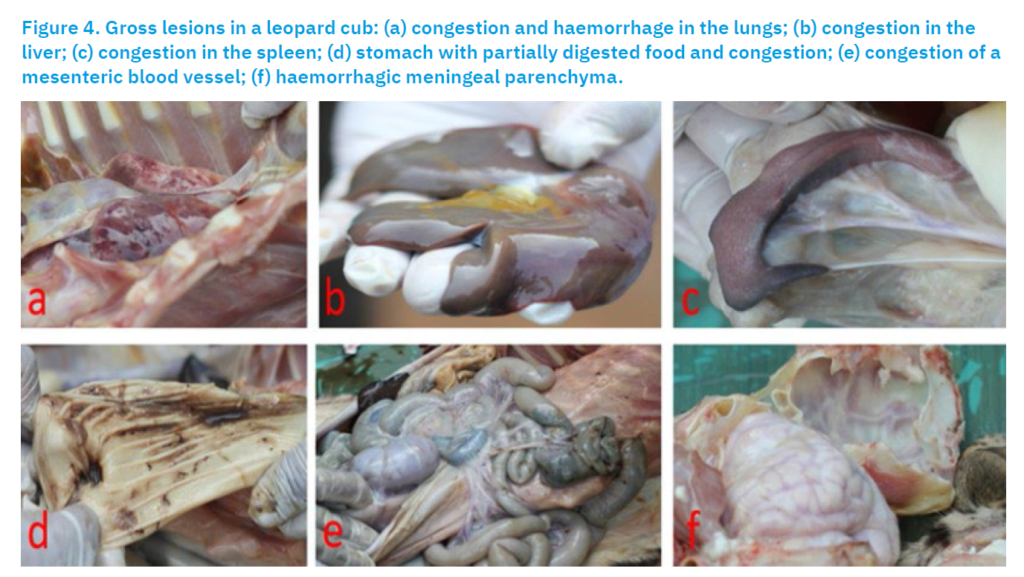
Necropsy of the deceased cub revealed congestion and haemorrhage in the lungs (Fig. 4a), liver (Fig. 4b), and spleen (Fig. 4c). The stomach contained partially digested food (Fig. 4d), and congestion was noted in the mesenteric artery (Fig. 4e). Additionally, haemorrhagic changes in the meningeal parenchyma (Fig. 4f) were observed in the brain.
In the laboratory, RNA was extracted from blood, swab samples (nasal and oral) and tissue homogenates (brain, lungs, and spleen) using RNAisoPlus® following Green and Sambrook (2012). The extracted RNA was reverse transcribed into cDNA using the Verso cDNA synthesis kit (Thermo Fisher Scientific, USA). Canine distemper virus was detected by nested PCR as described by Kim et al. (2001). The first round used outer primers (Outer F: 5′-ATTTGGGATTGCTTAGGA-3′; Outer R: 5′-GGCGCTCATCTTGGACAT-3′), followed by a second round with inner primers (Inner F: 5′-GTTAGCTAGTTTCATCCT-3′; Inner R: 5′-GGTCCTCTGTTGTCTTGG-3′). PCR reactions (10 µl) contained 5 µl 2× Master Mix (Ampliqon®), 1 µl each of forward and reverse primers (10 pmol), 1 µl cDNA, and 2 µl nuclease-free water. Thermal cycling was performed in a C1000 Bio-Rad thermal cycler with an initial denaturation at 94°C for 5 min, followed by 35 cycles of 94°C for 45 s, 54°C for 45 s, and 72°C for 45 s, and a final extension at 72°C for 7 min. Amplicons were resolved on 1.5% agarose gel at 75 V, sized using a 100 bp DNA ladder, and visualized under UV illumination.
Meanwhile, corneal and brain impression smears were examined for rabies using Seller’s staining and fluorescent antibody testing (FAT). Seller’s-stained smears were washed, air-dried, UV-exposed for 30 min, and examined under oil immersion for magenta Negri bodies (Young and Sellers, 1927). For FAT, brainstem smears were fixed in chilled absolute acetone for 2 h, rinsed in PBS (pH 7.2), and incubated with anti-rabies FITC conjugate (Merck Life Sciences Pvt. Ltd., Mumbai, India) at 37 °C for 30 min. Slides were washed thrice in PBS, mounted with aqueous mountant, and examined under a fluorescence microscope (Nikon ECLIPSE Ni, Japan) at 400× magnification; dusty apple-green fluorescence was considered positive. Rabid and rabies-free dog brain tissues served as positive and negative controls, respectively. Confirmation was performed by RT-PCR of corneal and brain tissue following Kadam et al. (2025).
Blood smears were initially examined to rule out blood protozoan infections, followed by PCR analysis for confirmation. Genomic DNA was extracted from blood samples using the Qiagen DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Multiplex PCR was performed using three-specific primers to detect Ehrlichia canis, Hepatozoon spp., and Babesia spp., as described by Tanasak et al. (2025).
For haematological analysis, 3 mL of blood was collected from the cephalic vein into heparinised vacutainers and analysed using an automated haematology analyser (Mindray BC-2800). Haemoglobin, total RBC and WBC counts, lymphocyte count, platelet count, and PCV were measured within 24 h of collection to prevent cell lysis. For serum biochemical analysis, blood samples (about 3 mL) were collected in EDTA-coated vacutainers, centrifuged at 3000 rpm for 15 min, and the serum stored at −20 °C until analysis. Serum total protein, albumin, glucose, creatinine, ALT, ALP, calcium, and phosphorus were estimated using commercial kits on an A15 Biosystems Automatic Biochemical Analyzer (Biosystems, S.A., Spain).
For toxicological analysis, (25 g each) of gut content and liver were processed at the Regional Forensic Laboratory, Tirunelveli. Samples, along with prepared controls, were placed in 500 mL plastic containers containing saturated brine solution, with 100 mL of preservative added prior to sample collection; sample weight was determined by difference. Residue analysis of seven pesticides—organochlorines (DDT, endosulfan), organophosphorus compounds (malathion, chlorpyrifos, fipronil), an organocarbamate (carbofuran), and a pyrethroid (cypermethrin)—was performed using a single quadrupole GC–MS system (Agilent) following the method of Octavio et al. (2014).
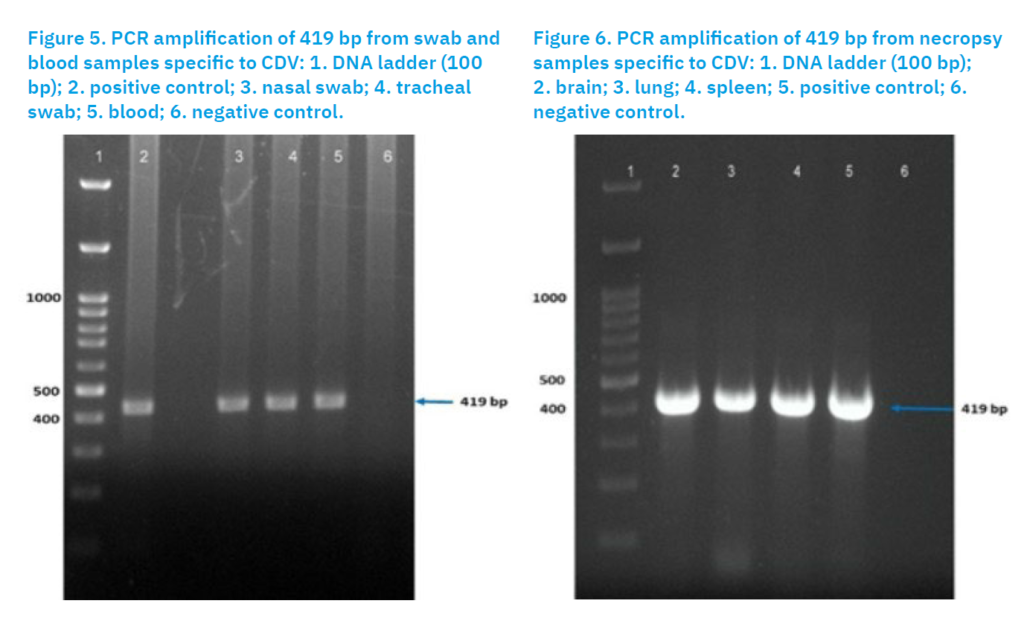
Laboratory findings indicated that PCR analysis of nasal and oral swabs and blood samples confirmed the presence of the CDV genome (Fig. 5). Similarly, screening of the collected tissue samples (brain, lung and spleen) also confirmed the presence of CDV (Fig. 6).
However, FAT and RT-PCR analyses of the corneal and brain samples tested negative for the rabies virus. Blood smear examination and multiplex PCR analysis of the blood sample were negative for blood protozoans. Haematological analysis revealed anaemia and lymphocytopenia, while biochemical tests indicated hypoglycaemia (Table 1). Pesticide analysis of seven compounds in the gut contents and liver samples was negative.
Treatment, outcome and follow-up
Treatment was initiated to manage the systemic and neurological signs of canine distemper in the leopard cub. Intravenous fluids (Fig. 7) were administered to correct dehydration and electrolyte imbalances, including Dextrose Normal Saline (100 ml I/V BID), Ringer’s Lactate (100 ml I/V BID), and 25% Dextrose (50 ml I/V OD) for the first two days. Broad-spectrum antibiotics—cefotaxime (400 mg, I/V BID) and metronidazole (150 mg, I/V BID)—were administered to control secondary bacterial infections.
Supportive therapy included mannitol (10 ml I/V OD) for suspected cerebral edema, prednisolone (10 mg I/M OD), Metaways (10 ml I/V OD for two days) for metabolic support, Astymin (10 ml I/V OD) as a multivitamin supplement, and Renerve Plus (2 ml I/V for five days) for neural repair. Oral supplements for anaemia and nutrition support (aRBCe, Thrombofit, AmiMeo, and Liv.52; each 5 ml BID) were provided, along with taurine (Taurians ½ tab BID). After five days, Siddha medicine was added, including Nilavembu Kasayam (3 ml TID), Poorna Chandrodaya powder (pinhead quantity in 10 ml NS OD), and Legiyam Prami Nei (one scoop BID). Fluid therapy was adjusted as required based on the clinical response.
The cub showed marked improvement following the introduction of Siddha medicine. By the tenth day, it was feeding independently, voiding normally, and exhibited no further convulsions or hypersalivation, with a noticeable improvement in body condition. However, by the fifteenth day, the cub became dull again, with reduced appetite and decreased urination and defecation. Despite continued treatment, its condition progressively worsened, and the cub died on the nineteenth day after presentation.
Discussion
Canine distemper virus (CDV) and related morbilliviruses infect a wide range of species, causing severe disease. Among carnivores, outbreaks have occurred in marine mammals such as seals, dolphins and porpoises (Barrett et al., 1993), and in large felids including lions, leopards, tigers and jaguars in North American zoos (Appel et al., 1994), as well as in tigers and leopards in Russia and Nepal (Konjević et al., 2011; Bodgener et al., 2023). Other susceptible carnivores include raccoon dogs and badgers (Kim et al., 1997; Hur et al., 1998), as well as grey wolves (Di Sabattino et al., 2014). CDV has also been detected in non-human primates such as Japanese, crab-eating, and rhesus macaques (Yoshikawa et al., 1989; Morikawa et al., 2008; Sun et al., 2010).
Leopards infected with canine distemper virus (CDV) exhibit clinical signs similar to those seen in domestic dogs, including hyperkeratosis of the footpads and ocular disease, as reported in a recent Javan leopard case (Rahman et al., 2022). Neurological signs are also common, and individuals showing severe neurological involvement often die (Bodgener et al., 2023). The present case likewise showed footpad hyperkeratosis and neurological symptoms, highlighting the severity and treatment challenges of CDV in wild leopards. Despite intensive therapy integrating modern veterinary and Siddha medicine, the cub ultimately succumbed to the infection.
Creevy (1986) noted that treatment of CD focuses on preventing secondary bacterial infections, maintaining hydration, and managing neurological signs. Supportive care may include antibiotics, fluids, nutritional support, antipyretics, analgesics, and anticonvulsants; however, no single therapy is consistently effective. While some animals recover with prompt, intensive treatment, outcomes are often poor once neurological symptoms develop, and severe progressive cases may warrant euthanasia. Some chronic neurological cases may respond to immunomodulatory therapies such as anti-inflammatories or steroids. In the present case, the leopard showed neurological signs and did not respond to treatment, dying after 19 days. A brief improvement following the integration of Siddha medicine suggests possible supportive benefits; however, controlled studies are needed to assess its efficacy.
Conclusion
This study describes the clinical presentation, diagnosis, treatment, and outcome of canine distemper in a leopard cub, representing the first documented case from southern India. However, sequencing of CDV-positive samples following conventional PCR could not be performed due to limited sample availability, which constitutes a limitation of the study.
Acknowledgement
The authors express their heartfelt gratitude to the Tamil Nadu Veterinary and Animal Sciences University for facilitating the publication of this research paper. The authors are also grateful to the District Forest Officers of Kanyakumari and Tirunelveli Divisions for the cooperation and encouragement extended in obtaining approval to publish the scientific data. Finally, due acknowledgement is extended to the Director of Ayush for the technical advice provided regarding Siddha treatment for the ailing animal.
References [… show]
Prvi dokazani slučaj virusa štenećaka kod mladunčeta leoparda (Panthera pardus) u Tamil Naduu u Indiji
Nakulan Valsala Rajesh1* (corresponding author), crocvet@gmail.com, orcid.org/0000-0003-0399-9051; Shunmugavel Manoharan2, smanovet@yahoo.com, orcid.org/0009-0005-7520-8281; Sundaram Muthukrishnan3, muthukrishnan.s@tanuvas.ac.in, orcid.org/0009-0004-2901-5342; Srinivasan Jaisree4, drjai81@yahoo.com, orcid.org/0000-0003-4962-620X; Panneerselvam Jayashree5, pjayashree2003@gmail.com, orcid.org/0009-0002-1888-8709; Elayaraman Madhesh1, madheshvmd@gmail.com, orcid.org/0000-0003-2137-9016; Radhakrishnan Bhuvaneshwari Vishnurahav6, vishnu.vet6@gmail.com, orcid.org/0000-0003-1962-4238, Muthusamy Ranjithkumar1, clmranjith@gmail.com, orcid.org/0000-0001-8389-4633; Rajagopalan Chandrasekaran Sundararajan1, rcsundararajan@gmail.com, 0000-0002-0843-1310; Elangovan Tamileniyan7 tamileniyan.2000@gmail.com, orcid.org/0009-0003-2459-9847.
1Department of Veterinary Medicine, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
2Forest Veterinary Assistant Surgeon, Forest Veterinary Unit, Forest Extension Division Campus, Ponnakudi, 627 151Tirunelveli, India.
3Department of Veterinary Anatomy, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
4Central University Laboratory, Centre for Animal Health Studies, Madhavaram Milk Colony, 600 051Chennai, India.
5UG Scholar, Veterinary College and Research Institute, 627 358Tirunelveli, India
6Department of Veterinary Physiology and Biochemistry, Veterinary College and Research Institute, 627 358 Tirunelveli, India.
7Internee, Veterinary College and Research Institute, 614 625 Orathanadu, India.Četveromjesečno mladunče leoparda (Panthera pardus) spašeno je iz stambenog područja u Kaliyalu, Kanyakumari, Tamil Nadu, Indija. Mladunče je pokazivalo znakove tuposti, konvulzivne napadaje i obilno slinjenje. Testirano je na zarazne bolesti, a virus štenećaka potvrđen je konvencionalnom PCR metodom. Koliko je poznato, ovo je prvi potvrđeni slučaj štenećaka kod leoparda u Tamil Naduu, Indija, a istraživanje je usmjereno na integrativni alopatski i siddha tretman.
Ključne riječi: alopatski tretman; štenećak; leopard; siddha tretman; Tamil Nadu

