Fusarium moulds, mycotoxins and their metabolites
M. Sokolovic*, A. Vulić, N. Kudumija, T. Lešić, J. Pleadin, M. Berendika and M. Zadravec
Marijana Sokolović1* (corresponding author), m_sokolovic@veinst.hr, orcid.org/0000-0003-3373-7415; Ana Vulić2, vulic@veinst.hr, orcid.org/0000-0002-9379-7236; Nina Kudumija2, kudumija@veinst.hr, orcid.org/0000-0001-6144-6514; Tina Lešić2, lesic@veinst.hr, orcid.org/0000-0001-6773-9473; Jelka Pleadin2, pleadin@veinst.hr, orcid.org/0000-0002-0768-0462; Marija Berendika1, mberendika@veinst.hr, orcid.org/0000-0002-6717-6292; Manuela Zadravec3, zadravec@veinst.hr.
1, Laboratory for Animal Feed Analytics, Poultry Centre, Croatian Veterinary Institute, 10000 Zagreb, Croatia
2 Laboratory for Analytical Chemistry, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
3 Laboratory for Feed Microbiology, Department of Veterinary Public Health, Croatian Veterinary Institute, 10000 Zagreb, Croatia
https://doi.org/10.46419/cvj.57.4.6
Abstract
Moulds of the genus Fusarium are present in a range of aquatic and soil environments as pathogens, endophytes and/or saprophytes. They have ability to easily adapt to different environmental conditions, enabling the ease of their spread worldwide. However, their growth and development occur more often in the areas with a humid and warm climate. The majority of Fusarium species are plant pathogens, but certain strains can cause diseases in humans and animals either as direct pathogens or by indirect action of their mycotoxin metabolites. They can cause acute and chronic intoxication and disrupt the function of various organ systems. The most important groups of Fusarium mycotoxins are the trichothecenes, fumonisins and zearalenones. Modern technologies, instruments and methods have enabled discovery of a large number of Fusarium moulds and their metabolites, though toxigenic functions have been described only for a small number of species. For most newly discovered metabolites, there is no detailed information on their structural and chemical characteristics, mode of biosynthesis or evidence of their potential (un)desirable bioactive effects. Considering the unavoidable common presence of Fusarium moulds and/or their metabolites, as well as their potential harmful effects, further research on newly discovered species and metabolites is necessary. The aim of this review paper is to provide an overview of the current knowledge on Fusarium species and their metabolites, with a focus on their growth characteristics and metabolite biosynthesis.
Keywords: Fusarium, moulds, mycotoxins, metabolites, toxicity.
Introduction
Fusarium moulds are ubiquitous worldwide as endophytes, saprophytes and/or pathogens of plants, humans and animals. Infection of plants depends upon pathogenicity of the pathogen, plant resistance and environmental factors (e.g., temperature and humidity), resulting in symptoms of disease and often plant death, causing significant economic losses in agriculture. The most common pathogenic Fusarium species that cause plant diseases are F. graminearum, F. oxysporum, F. sporotrichioides and F. verticillioides. These species have enormous biosynthetic potential and produce a range of primary and secondary metabolites (enzymes, proteins and (myco)toxins). Currently, more than 700 Fusarium metabolites have been described (Crous et al., 2021).
Fusarium mycotoxins are secondary metabolites of different chemical structures. They are considered hazardous to human and animal health due to their potential harmful effects (e.g., oestrogenic, haemorrhagic, hepatotoxic, immunomodulatory, carcinogenic, mutagenic, neurotoxic and teratogenic). The most commonly detected mycotoxins in food and feed are trichothecenes (TCTs), fumonisins (FUMs) and zearalenones (ZENs) (Desjardins, 2006; Eskola et al., 2020; Kolawole et al., 2024).
Application of molecular principles in mould identification and modern analytical methods and instruments in the detection of their metabolites has significantly enhanced this area of research. The use of molecular methods resulted in identification of numerous Fusarium (toxigenic) species (Leslie and Summerell, 2006; Winter et al., 2018; Naeem et al., 2019). Similarly, application of multi-toxin analytical methods, in addition to the commonly detected diacetoxyscirpenol (DAS), deoxynivalenol (DON), T-2/HT-2 toxins, nivalenol (NIV), ZENs and FUMs, enabled detection of newly discovered and potentially toxic metabolites, their derivatives and conjugates (Roush et al., 1985; Avila et al., 2019; Villani et al., 2019; Qiu et al., 2020; Vulić et al., 2021). There is a great body of literature data on the incidence and toxicity of Fusarium species and their metabolites. However, use of different identification protocols and toxicity analyses hinders their comparisons. In respect to the high occurrence of these moulds and their potential toxicity, this paper provides an overview of known Fusarium species, focusing on their growth and development and the biosynthesis of their associated mycotoxins and metabolites.
Genus Fusarium
The name Fusarium was pioneered by Link in 1809, while the toxic secondary metabolites were identified much later, in the 1970s. The majority of literature data on Fusarium moulds are based on identification protocols described by Wollenweber and Reinking (1935), Snyder and Hansen (1940), Booth (1971), Gerlach and Nirenberg (1982), and Nelson, Tousson and Marasas (1983). These were based on morphological characteristics, while the results significantly differed in respect to the number of defined species and varieties (Nelson, 1994). The morphological, physiological, and ecological diversity of Fusarium species, and their ability to quickly adapt to adverse environmental conditions are specific features that make the identification and classification cumbersome. Innovative approaches using molecular methods in mould identification have enabled accurate identification and classification of isolated strains and phylogenetic verification of data from previous research studies (O’Donnell et al., 2009, 2012; Schoch et al., 2020; Lešić et al., 2024).
According to the currently accepted taxonomy as described in the NCBI database (National Center for Biotechnology Information), the genus Fusarium belongs to the family Nectriaceae, order Hypocreales, class Sordariomycetes, phylum Ascomycota and subkingdom Dikarya of the kingdom Fungi. More than 300 phylogenetically distinct species are known, although not all have been analysed in detail (Jacobs-Venter et al., 2018; Schoch et al., 2020). Since 2011, the system of only one name of the genus Fusarium has been accepted, i.e., different names for asexual (anamorphic) and sexual (telomorphic) phases are no longer used (Geiser et al., 2004). The morphological classification generally includes analysis of the macroscopic characteristics of the colony growth on nutrient agars (e.g., shape, size, texture and colour of the surface and back of the selective nutrient agar) and microscopic characteristics (e.g., conidia morphology, morphology and presence of septa and surface of ascospore, morphology of telomorphic forms, etc.) (Moretti et al., 2017; Crous et al., 2021; Lešić et al., 2024).
A great number of described species are saprophytic soil moulds and plant pathogens. Only a small number of them is considered as pathogens for humans and animals. Certain species more often infect specific hosts (wheat, corn, soybeans, sugar beets, sunflowers, potatoes, bananas, various vegetables and fruits and other plants), causing characteristic symptoms of diseases (e.g., head blight, dry rot, root and stem rots, wilt, necrosis, sudden death syndrome of soybean, etc.) (Summerell et al, 2003; O’Donnell et al., 2012; Johns et al., 2022). Although a detailed description of these changes is beyond the scope of this review paper, they should be pointed out, given the enormous economic losses in agriculture, but also to elucidate the toxicogenic potential of these pathogens. According to occurrence reports of moulds and their toxic metabolites in crop production, the most common species are F. graminearum, F. culmorum, F. langsethiae, F. proliferatum, F. sporotrichioides and F. verticillioides. Mycotoxins contaminate 60-80% of cereal grains and in 20% of these contaminations, the concentration of mycotoxins exceeds the levels indicated in regulations and guidelines of the European Commission (EC) (Eskola et al., 2020; Johns et al., 2022). The incidence of certain species in different geographical areas is constantly changing, indicating the need for precise identification of pathogenic strains (JECFA, 2001; Pleadin et al., 2023). In addition to accurate morphological and genotypic identification of isolated moulds, assessment of their potential for infection of (different) hosts, evaluation of disease symptoms, and research on their toxicogenic potential is of high importance.
Toxigenicity of Fusarium species
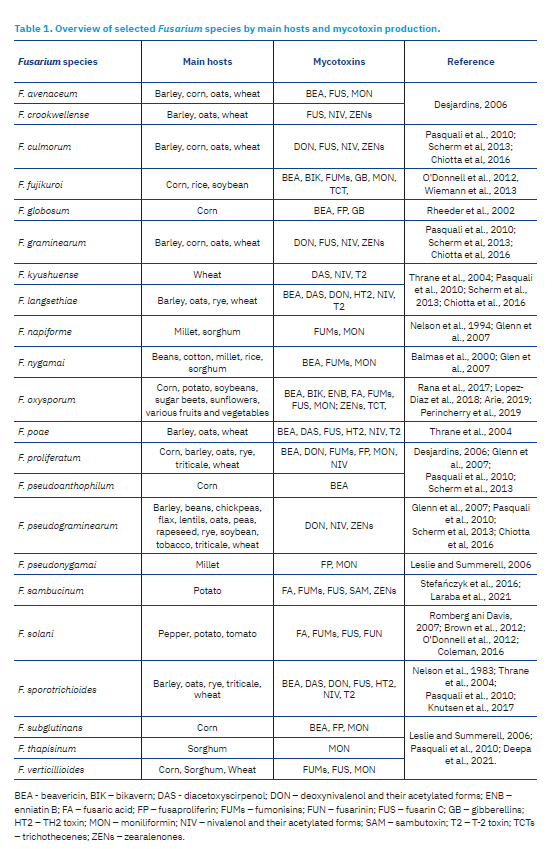
Toxigenicity has been confirmed for a number of isolated strains of many species of Fusarium. For example, isolated strains of F. graminearum, F. pseudograminearum and F. culmorum cause fusarium head blight in wheat, barley, oats and triticale, and foot and root rot in wheat, barley, oats, rye, triticale, flax, rapeseed, soybean, tobacco, chickpeas, lentils, beans and peas, and produce Type B trichothecenes (DON, acetylated derivatives 3- and 15-acetyl-deoxynivalenol (3-ADON, 15-ADON) and NIV) (Pasquali et al., 2010; Scherm et al., 2013; Chiotta et al., 2016). F. oxysporum cause wilt of soybeans, sugar beets and sunflowers, but can also cause tuber rot in potato and various fruits and vegetables (including banana, watermelon, cantaloupe, tomato, spinach, pea and cucumber), while in corn they cause root, stem, ear and seed rot. During infection, these species often produce mycotoxins in plants before harvest or later during storage. The most commonly produced mycotoxins are TCTs, FUMs, fusarin, fusaric acid (FA), beauvericin, enniatin B, bikaverin, moniliformin (MON) and ZENs (Rana et al., 2017; Lopez-Diaz et al., 2018; Arie, 2019; Perincherry et al., 2019). F. fujikuroi cause fusariosis (wilt and plant rot) of rice, corn, soybean, and also produce TCTs, fusarin, FUMs and bikaverin (O’Donnell et al., 2012, Wiemann et al., 2013). F. sambucinum is a causative agent of potato fusariosis (root rot) and it also produces sambutoxin (SAM), FA, FUMs, ZENs and fusarin C (Stefańczyk et al., 2016; Laraba et al., 2021). F. solani infects potato, tomato and pepper with symptoms of fusariosis and produces FAs, FUMs, fusarin and fusarinin (Romberg and Davis, 2007; Brown et al., 2012; Coleman, 2016). An example of the toxicogenic potential of certain mould species is shown in Table 1. This toxigenicity depends not only on the mould species, but also on the host plant and environmental conditions (Glenn et al., 2007).
Accordingly, differentiating beneficial saprophytic organisms from potentially harmful ones, and their precise identification, can be achieved by morphological analysis (identification to the genus level), phylogenetic analysis (identification to the species level), determination of host-specific toxic effects, and their ability to produce (toxic) secondary metabolites (Hofstetter et al., 2019; Summerell, 2019; Schoch et al., 2020).
Fusarium life cycle and mycotoxin production
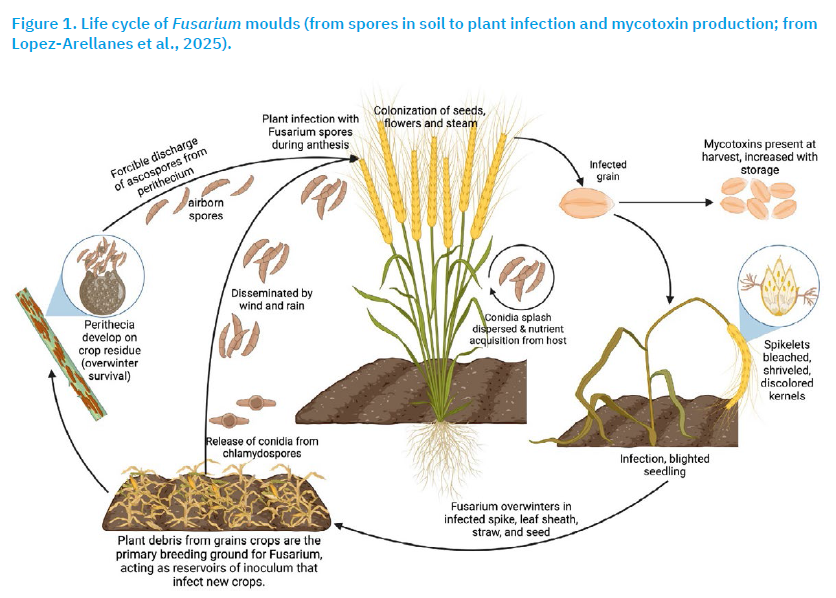
Fusarium species are commonly called field moulds because fusariosis of plants are often caused by spores in the soil that are present in the plant debris (Summerell, 2019). Due to their ability for rapid morphological and physiological adaptation to diverse conditions, they can survive for long periods even in unfavourable conditions. For example, they can survive in the soil for more than ten years as chlamydospores, as mycelium on plant debris or as saprophytes (on dead organic matter). While some species are present in all climates, other species prefer tropical, dry and hot and/or temperate climates (Summerell et al., 2003; Verheecke-Vaessen et al., 2019; Oluwakayode et al., 2024).
Spores are easily spread in the environment by wind and rain (but also with plants, animals and soil) and find new hosts. Under favourable conditions (e.g., adequate temperature and humidity of air and soil, pH of soil, etc.) and other growth stimulating factors (damage and stress of plants), the spores (asexual or sexual from perithecia) invade a new host (reaching root, stem, leaves, grain and fruit of the plant) and develop either sexually or asexually forming sexual-ascospore or asexual microconidia, macroconidia and chlamydospore (Figure 1). It has been estimated that only 20% of Fusarium species has a sexual growth phase (called the telomorphic phase) (Ma et al., 2013). During growth, moulds produce primary and secondary metabolites. Primary metabolites are essential for growth and development. On the other hand, secondary metabolites are not necessary for survival, but can enhance pathogenicity and virulence. Depending on the structure, absorption and biotransformation processes, certain metabolites can also have specific bioactive effects (e.g., antifungal, cytotoxic, phytotoxic) (Summerell et al., 2003, 2019; Schoch et al., 2020; Lopez-Arellanes et al., 2025).
Fusarium mycotoxins
Mycotoxins are secondary metabolites of low molecular weight (<2000 Da) that are produced by moulds during their secondary growth phase. Like the other secondary metabolites, they are not essential for the growth and development of moulds, but they contribute to their pathogenicity by preventing the growth of competitive microorganisms on the same substrate or by helping to invade host cells. Mycotoxin production can occur in the mould itself, in infected plants at all stages of plant growth and development (before and after harvest) but also during plant processing and storage and in the organisms of humans and animals. In most cases, one mould simultaneously produces different mycotoxins and these metabolites combinations are often found during chemical analysis of food and feed (Berthiller et al., 2015; Crous et al., 2021; Kolawole et al., 2024).
The quantity and type of produced toxins and metabolites depend upon many factors, including physical factors (e.g., moisture, relative air humidity, temperature, mechanical damage), chemical factors (e.g., oxygen and carbon dioxide content, substrate composition), and biological factors (e.g., plant species, plant health, quantity of spores). Once produced, mycotoxins are very stable compounds and are not easily deactivated by standard food and feed processing procedures (O’Donnell et al., 2012; Gil-Serna et al., 2014; Bryla et al., 2018; López-Arellanes et al., 2025). It is well known that Fusarium mycotoxins can cause harmful effects in human, animals and plants. For certain mycotoxins, the link between contaminated food consumption and the onset of disease symptoms (immunomodulatory, haemorrhagic, carcinogenic, hepatotoxic and neurotoxic effects) in humans and animals has been established and verified. Furthermore, they cause lower production results in animal industry (reduced feed intake, feed refusal, lower feed conversion ratio, lower reproducibility and increased tendency to other diseases (Desjardins, 2006; Li et al., 2020, 2025). Although a great number of modified forms and newly discovered mycotoxins have been described in the scientific literature, mycotoxicosis has been described for a rather small number of toxins (e.g., TCTs, FUMs and ZENs).
Trichothecenes (TCT) are a large group of metabolites that share the same basic chemical structure characterised by the presence of a tetracyclic sesquiterpenoid (e.g., sesquiterpenoid 12,13-epoxytrichothec-9-ene). More than 190 metabolites have been described so far, and their name originates form the first isolated metabolite (trichothecin). According to their structural characteristics, they can be grouped as macrocyclic (molecules with a ring) and non-macrocyclic (molecules without the ring between C-4 and C-15) toxins. They are further divided into Types A, B, C and D. The most prominent representatives of the tricoherences are T-2 toxin, HT-2 toxin, DAS and neosolaniol (NEO) of Type A toxins and DON and NIV that are classified as Type B toxins. Macrocyclic TCTs of Types C and D are verrucarins, roridins, satratoxins and baccharins. In general, they are considered more toxic than Type A and B toxins, but also less hazardous due to their relatively rare occurrence in nature. Since they are not commonly analysed in the samples, they are also insufficiently studied (Leeson et al., 1995; PubChem, 2025).
Production of metabolites and biotransformation of toxin molecules can occur in moulds, plants, animals, and humans. They differ in respect to the substrate (e.g., plant species, plant parts, soil characteristics, metabolism, presence and activity of other microorganisms, etc). In general, the process includes phase I reactions (oxidation, reduction and hydrolysis) and phase II reactions (conjugation). Primary phase I reactions result in the production of molecules of similar toxicity, while further modifications generate significantly fewer toxic metabolites. Many harmful effects of TCTs have been described, the most important of which are inhibition of DNA, RNA and protein synthesis, damage and malfunction of digestive, circulatory, immune and nervous system (Leeson et al., 1995; Sokolovic et al., 2008; Nathanail et al., 2015; Knutsen et al., 2017).
T-2 toxin (fusariotoxin, insariotoxin, C24H34O9) and HT-2 (C22H32O8) toxin are low molecular weight molecules (466 g/mol and 424 g/mol), classified as Type A non-macrocyclic TCT toxins. They are weakly soluble in water and petroleum ether, but soluble in ethanol, ethyl acetate, chloroform, dimethyl sulfoxide and other organic solvents. They are produced by F. equiseti, F. graminearum, F. langsethiae, F. oxysporum, F. poae, F. roseum, F. sambucinum, F. solani, F. sporotrichoides, F. sulfureum, F. tricinctum and other Fusarium species, as well as moulds from the genera Cephalosporium, Myrothecium, Stachybotrys, Trichoderma, Trichothecium and Verticimonosporium (Krska et al., 2001; Nielsen and Thrane, 2001; Sokolovic et al., 2008; EFSA, 2017a, PubChem, 2025).
Biotransformation of T-2 toxin in moulds, plants and animals include reactions of both phase I and II. During phase I, reactions of hydrolysis, oxidation and reduction modify T-2 toxin into HT-2 toxin, NEO, T2-triol and more than 20 other modified forms. T-2 toxin and their phase I metabolites can form conjugates with polar molecules (carbohydrates and sulphates) during phase II. In plants, conjugates are formed mainly with glucose or sulphate, while in animals, conjugates are formed with glucuronic acid. Chromatographic analysis of chemical composition and identification according to their mass-to-charge ratio using mass spectrometry has enabled the detection of more than twenty conjugates. However, only a small number of them have been synthesised so far (e.g., T2-3-Glc, T2-Sulf, 3-Ac-T2). A noteworthy fact is that T2-3-Glc, as the primary conjugate of T-2 and other modified forms, has also been isolated from plants (McCormick et al., 2012, 2015; Wu et al., 2014; Nathanail et al., 2015, 2016; EFSA, 2017a). In animals, after absorption through the skin or digestive tract, T-2 toxin quickly arrives in the bloodstream, is hydrolysed into HT-2 and distributed to the liver, kidneys and other organs (without accumulation). Although not sufficiently confirmed, it is assumed that the phase II metabolites are formed in the digestive system (conjugation with glucuronic acid of sulphate) and that all metabolites (from phase I and II) along with the parent molecule T-2 (or HT-2) toxin are excreted out of the body within three days via urine or faeces (EFSA, 2017a; Freire et al., 2018).
The toxic effect of T-2 toxin in humans is the outcome of a sequence of activities of parent molecules and their derivatives (all metabolites that are formed in the mould, infected plants and other organisms). The impact of modified forms in respect of their toxicity still needs to be clarified. However, the main culprit for T-2 toxicity is the structural epoxide (C12-C13). Since it is found in both the parent molecules and their modified forms, it can be assumed that they also possess substantial toxicity. Further research will elucidate the extent of their toxicity. By evaluation of toxicological studies and data, the tolerable daily intake (TDI) in food for T2 and HT2 toxins is set at 0.2 µg/kg body weight. Such a low level of these substances in food that can be safely ingested over a lifetime without undesirable health effects is set because of the confirmed immune and hepatotoxic effects, inhibition of DNA, RNA and protein synthesis, induction of apoptosis and lipid peroxidation. In humans, T2 and HT2 intoxication has been described as alimentary toxic aleukia (impairment of the immune systems, haemorrhage, necrotic lesions, pancytopenia and sepsis). Similar symptoms have also been described in animals. It is important to notice that in ruminants, due to the specificity of their digestive system and the presence of rumen microflora and consequent toxin metabolism, the observed toxic effects are less pronounced (Desjardins and Plattner, 1989; Nathanail et al., 2015; EFSA, 2017a).
Diacetoxyscirpenol (DAS, Anguidine, 4,15-Diacetoxyscirpenol, C19H26O7) is considered a highly toxic TCT with a molecular weight of 366.4 g/mol. It is stable when exposed to heat and UV radiation, and soluble in moderately polar solvents (acetone, diethyl ether, and ethyl acetate). Several species of Fusarium moulds are producers, including F. acuminatum, F. avenaceum, F. clamydosporum, F. compactum, F. crookwellense, F. culmorum, F. equiseti, F. graminearum, F. langsethiae, F. moniliforme, F. poae, F. roseum, F. sambucinum, F. semitectum, F. solani, F. sporotrichioides, F. sulphureum, F. tricinctum and F. venenatum (Jelen et al., 1995; Leeson et al., 1995; Nielsen and Thrane, 2001; McCormick et al., 2011; Schollenberger et al., 2011; PubChem, 2025).
The process of biotransformation, as for other molecules, differs depending on the site where the process takes place. Research on moulds (F. verticillioides) has shown that 3,15-DAS and other deacylated products are made by acetylation and deacetylation. In animals, this compound is also metabolised in two phases. During phase I, hydrolysis and hydroxylation reactions produce NEO, 8ß-OH-DAS, 7-OH-DAS, 4-MAS, 15-MAS and scirpentriol (SCP). In the phase II, the metabolites DAS-3 glucuronide, 15-MAS 3-glucuronide and 15-MAS-4 glucuronide are produced (Roush et al., 1985; Yang et al., 2015; EFSA, 2018a). For example, during biotransformation of DAS in corn samples, 15-MAS and 4,15-DAS are modified into the glucosides 15-MAS 3-glucoside and 4,15-DAS 3-glucoside, respectively (Schollenberger et al., 2011). However, in the research on biotransformation in rats, it has been demonstrated that 4,15-DAS is modified into 4,15-DAS 3-glucuronide (Roush et al., 1985). In addition to these modifications, during metabolism in plants, due to microbial transformation or thermal reactions, a number of acetylated and hydroxylated derivatives may be produced, including 7,8-dihydroxydiacetoxyscirpenol (7,8-diHDAS), 7-hydroxydiacetoxyscirpenol (7-HDAS), 4,15-diacetylnivalenol (4,15-diANIV) and 4β,8α,15-triacetoxy-3α,7α-dihydroxy-12,13-epoxytrichothec-9-ene), and various glucosides and glucuronides (Garies et al., 1990; Nakagawa et al., 2013; Escrivá et al., 2017; Yoshinari et al., 2018; Li et al, 2020).
Literature data on the occurrence and toxicity of DAS are scarce. However, using the available data and the similarity of this toxin to T2 toxin, the estimated TDI value for DAS is around 0.06 µg/kg body weight for humans. Sublethal concentrations of these toxins can cause damage to organs of the immune, lymphatic and digestive systems, while symptoms of intoxication in humans and animals include vomiting, diarrhoea, blood pressure drop and suppression of the immune system. A number of adverse health effects of this toxin have been described in poultry, pigs, ruminants, dogs, cats, fish and other animals. As other TCTs, when ingested, the toxin can cause vomiting, diarrhoea and lethargy in animals, in addition to disruptions of the digestive, circulatory and immune systems (Leeson et al., 1995; EFSA, 2018a).
Deoxynivalenol (vomitoxin, DON, C15H20O6) is one of the most commonly reported TCTs in cereals. The molecular mass of DON is 296.3 g/mol. The molecule is soluble in polar organic solvents (e.g., aqueous solutions of acetonitrile, ethanol, methanol, chloroform, ethyl acetate) and in water. Like other TCTs, it is also resistant to heat and UV radiation. Among Fusarium species, the most common producers are F. acuminatum. F. crookwellense, F. culmorum, F. sporotrichioides, F. graminearum, F. poae, F. sambucinum, F. solani, F. sulphureum and F. tricinctum (Burlakoti et al., 2008; EFSA, 2017b; PubChem, 2025).
DON biotransformation has been described in detail in plants. The application of modern chromatographic methods and research on their modification resulted in detection of a great number of metabolites: DON-3-glucoside (DON-3Glc), DON-Hexitol, DON-S-cysteine, DON-S cysteinyl-glycine, DON-glutathione, DON-di-hexoside, DON-2H-glutathione, DON-malonyl-glucoside, 15-acetyl DON-3-glucoside, 3-Acetyl-DON, DON-3-sulfate, DON-15 sulphate, 3-epimer-DON, norDON A, B and C, norDON-3 glucoside A, B, C and D; DON-3-glucoside-lactone; de-epoxy DON, DON-glucuronide, de-epoxy DON-3-sulfate and de-epoxy DON-15-sulfate. Among these, the most commonly detected modified forms in food are 3-Ac-DON and 15-Ac-DON, in addition to certain glucosides and glucuronides. The well known conjugate DON-3Glc is the main product of the detoxification process and is therefore significantly less toxic to plants. It is interesting to notice that there is a link between the DON-3Glc/DON ratio and the susceptibility of cereals to Fusarium head blight, which has been detected in wheat, corn, barley and other cereals. Despite its lesser toxicity in plants, the problem occurs when it is ingested by humans and animals. Namely, after ingestion, it can be easily metabolized into more toxic forms (mainly through hydrolysis reactions). Generally, in animals and humans, DON is absorbed slowly, is metabolised in the digestive system and excreted from the organisms like other TCTs (Berthiller et al., 2005, 2015; Ovando-Martinez et al., 2013; Gratz et al., 2017; Freire et al., 2018).
Toxicity of DON and its derivatives in animals is characterised by the disruption of digestive function (e.g., reduced feed intake or refusal, vomiting and other digestive disorders). The most sensitive domestic animals are pigs, while these toxic effects are less pronounced in poultry and ruminants. DON is considered as the most important representative of TCT Type B due to its high occurrence, often in concentrations that induce toxic effects (EFSA, 2017b; Döll and Dänicke, 2004, 2011; Dänicke and Brezina, 2013). For example, 2 mg/kg DON in swine feed causes feed refusal and weight loss, while in poultry these symptoms are observed at significantly higher concentrations of toxins in the feed. Nonetheless, food and feed rarely contain only one mycotoxin. Hence, even a low concentration, in the presence of other mycotoxins, DON can have adverse health effects (JECFA, 2001; Bryden, 2012; Pinton et al., 2012; Bryla et al., 2018). Therefore, detection of DON, 3-Ac-DON, 15-Ac-DON and/or DON-3Glc in food and feed is considered equally dangerous because these modified forms can be easily metabolised in DON (Berthiller et al., 2011). Furthermore, although they are considered equally toxic, research studies have confirmed that DON and 15-Ac DON are most toxic due to their cytotoxic effects and inhibition of the protective function of the intestinal epithelium. The estimated TDI is 8 μg/kg body weight for humans (Nagl et al., 2014; Alizadeh et al., 2016; EFSA, 2017b).
Nivalenol (NIV, 12,13-Epoxy-3,4,7,15-tetrahydroxytrichothec-9-en-8-one, C15H20O7) is a mycotoxin classified as a Type B trichothecene, whose chemical structure is similar to DON (differing only in one oxygen atom at the position C-4). The NIV molecule, with a mass of 312.3 g/mol is stable and soluble in organic solvents. It is mostly produced by F. asiaticum, F. cerealis, F. cortaderae, F. crookwellence, F. culmorum F. graminearum, F. nivale, F. poae, F. tricinctum and other Fusarium species, and by species in the genera Stachybotrys and Trichoderma (Pettersson, 1991; Hope and Magan, 2003; PubChem, 2025). It can be found in different substrates including corn, wheat, oat, rice, peanuts, various legumes and other plants. It is produced in a wide range of temperatures (from 10–35°C) and water activities (from 0.99 aw to 0.96 aw) (Magan et al., 2010; Gomes et al., 2015; Pasquali et al., 2016; Moretti et al., 2017; Nazari et al., 2018; Vandicke et al., 2019). NIV is produced by a series of enzymatic reactions during the biotransformation process of 3,15-acetyl-DON and 3,4,15-triacetoxyscirpenol (metabolite of T-2 toxin) (Kumar et al., 2022; PubChem, 2025).
Toxic effects occur as a result of inhibition of DNA and protein synthesis and subsequent impairment of digestive function (e.g., damage of the intestinal epithelial cells, haemorrhage, congestion and diarrhoea), immune system (e.g., damaged bone marrow, spleen and thymus), induction of emesis and haemorrhage in the lungs and brain. In humans, such changes are described in idiopathic spastic paraparesis. Unlike DON, NIV is highly cytotoxic, inhibiting cell proliferation and causing oxidative stress. Considering its confirmed toxic effects and relatively common occurrence in cereals and human food at concentrations up to 107.2 μg/kg, the estimated TDI value of NIV is 1.2 μg/kg body weight (EFSA, 2013, 2017b; Kumar et al., 2022).
Fumonisins (FUM) are polyketides, classified in groups A, B, C and P. So far, 28 structural analogues have been described: FA1, FA2, FA3, PHFA3a, PHFA3b, HFA3, FAK1, FBK1, FB1, Iso-FB1, PHFB1a, PHFB1b, HFB1, FB2, FB3, FB4, FB5, FC1, N-acetyl-FC1, Iso-FC1, N-acetyl-iso-FC1, OH-FC1, N-acetyl-OHFC1, FC3, FC4, FP1, FP2, and FP3. Among these, the most important compounds are those from the group B (FB), namely, fumonisin B1 (FB1, C34H59NO15), fumonisin B2 (FB2, C34H59NO14) and fumonisin B3 (FB3, C34H59NO14), since they are frequent natural food contaminants. FB1 has a molecular weight of 721.7 g/mol, while FB2 and FB3 have molecular weights of 705.8 g/mol, and all are soluble in methanol. Furthermore, FB1 is considered the most toxic (Rheeder et al., 2002; PubChem, 2025). The most common producers of FUMs are F. fujikuroi, F. globosum, F. moniliforme, F. nygamai, F. proliferatum, F. subglutinans and F. veerticillioides, and the species Aspergillus niger and Alternaria alternata (Rheeder et al., 2002; Kamle et al., 2019; Anumudu et al., 2025).
Biotransformation of the parent molecules of FB occurs in the animal, and/or plant organisms, and produced are hydrolysed or partially hydrolysed compounds. Up to the present moment, application of different chromatographic methods on food, feed and other substrates enabled detection of FBs (FB1, FB2 and FB3), partially hydrolysed FBs (pHFB1a pHFB1b, pHFB2a, pHFB2b, pHFB3a and pHFB3c) and hydrolysed FBs (HFB1, HFB2 and HFB3). The process of biotransformation of FBs and their hydrolysed forms has been confirmed in swine and poultry, and these compounds are considered potential biomarkers of FB exposure (Zhang et al., 2024).
Furthermore, FB1 is cytotoxic and carcinogenic (e.g., causes carcinoma of the liver and oesophagus in humans), while in animals it causes disruption of the nervous and cardiovascular systems and damage of the lungs, kidneys and liver. Since carcinogenic activity of this toxin is confirmed in humans, it is classified as a 2B carcinogen. In animals, alimentary toxicoses are described, such as leukoencephalomalacia in horses (in concentration of 1–126 mg/kg of feed), pulmonary oedema in pigs (1–330 mg/kg in feed) and symptoms of diseases in poultry (feed contaminated with 80 mg/kg or more) (Gelderblom et al., 1984; IARC, 2002; Rheeder et al. 2022). The mechanism of toxic action is presumably linked to their structural similarity with sphingolipid bases. Namely, when an adequate concentration of FBs is ingested, this results in inhibition of sphingolipid metabolism, accumulation of intermediate compounds in the serum, and a decrease in the proportion of the complex sphingolipids that are essential for physiological functioning of the central and peripheral nervous system (Soriano et al., 2005; Kamle et al., 2019; EFSA 2022).
Zearalenone (Zenon, F-2 mycotoxin, ZEA, ZON, F-2, C18H22O5) is a nonsteroidal oestrogen that structurally resembles oestrogens (oestradiol, oestrone, oestriol, 7-beta-oestradiol and 17-beta-oestradiol). It is sometimes called phytoestrogen. This compound of molar mass of 318,4 g/mol is thermostable, although not in the presence of oxygen. It is soluble in alkaline aqueous solutions and fats, less soluble in acetone, acetonitrile, benzene, dichloromethane and methanol, while nearly insoluble in hexane and water (Bennet and Klich, 2003; PubChem, 2025). They are produced by species F. acuminatum, F. cerealis, F. crookwellense, F. culmorum, F. equiseti, F. graminearum, F. nivalis, F. oxysporum, F. semitectum, F. sporotrichioides and F. verticillioides in a wide range of temperatures (from 8–25°C) and with humidity above 20% (Gil-Serna et al., 2014; Kovalsky et al., 2014; Medina et al., 2017; Haque et al., 2020; Deepa et al., 2021; Tiru et al., 2022).
ZON is biosynthesized through a series of enzyme catalysed chemical reactions of a polyketide pathway in which two isomers (trans and cis) are produced. In plants, it is modified through the formation of glucose conjugates and ZON-14-o-beta-glucoside. In animals, it is metabolized in the intestinal cells where two main metabolites are formed: alpha-zearalenol and beta-zearalenol (ZEL metabolites are formed by the reduction reaction of the basic molecule, phase I reactions). Additionally, in phase II reactions, ZAL metabolites are formed: alpha-zearalanol and beta-zearalanol. In addition to these metabolites, a number of conjugates with glucuronic acid have also been described. Modifications of the original molecule significantly affect the bioactivity of metabolites, and certain forms are more common in different animal species. For example, alpha-ZEL has a stronger oestrogenic activity (i.e., activation process) in comparison to alpha-ZAL and ZON. This metabolite is mainly formed in the intestinal cells of swine and often causes disease. On the contrary, in the intestinal cells of poultry and ruminants, a deactivation process predominates during which beta-ZAL is formed, and it has weaker oestrogenic activity. This fact presumably explains the lower toxic effects of ZON in these animal species (Gareis et al., 1990; Dänicke et al., 2005; Minervini et al., 2006; EFSA, 2017c: Rai et al., 2019, 2020; Ropejko and Twaružek, 2021).
According to available data in the literature on toxicity, ZON has immunotoxic, hepatotoxic, xenogenic, anabolic and oestrogenic effects and its activity depends upon the immune status, age and sex of the animal. In general, ZON toxicity is highest in swine (at concentrations of 1–5 mg/kg in feed) and lowest in poultry (Leeson et al., 1995; Takemura et al., 2007; Minervini and Dell’Aquila, 2008). Acute and chronic intoxication with ZON in swine results in reproductive disorders (reduced fertility and litter size, abortions) and the development of typical clinical symptoms related to hypoestrogenism (vaginal and rectal prolapse, swollen mammary glands, foetal resorption and testicular atrophy). Considering that ZON and its metabolites can be found in food (mostly of plant origin) and given the potential toxic effects (oestrogenic, immunotoxic and carcinogenic), the estimated TDI value is set at 0.25 μg/kg body weight (Kuiper-Goodman et al., 1987; JECFA, 2001; EFSA, 2017c; Ropejko and Twaružek, 2021).
Fusaric acid (FA, 5-butyl-picolinic acid, C10H13NO2) was described in 1937 as a secondary metabolite of the mould F. heterosporum (Yabuta et al., 1937). For a long time, FA was considered a phytotoxin (mainly in rice) with negligible toxicity towards humans and animals (Niehaus et al., 2014; López-Díaz et al., 2018). It is a metabolite with a molecular mass of 179.2 g/mol, soluble in water and organic solvents (e.g., ethanol, methanol and DMSO) and by its chemical structure, it is classified in the pyridines. It is produced by the Fusarium species: F. fujikori, F. heterosporum, F. moniliforme, F. oxysporum, F. sacchari, F. solani, F. verticillioides, and others. In respect to their occurrence in Fusarium species, this compound has been used as a biomarker of contamination of rice and other cereals with these moulds.
The process of biotransformation has not been fully elucidated, although it has been confirmed that under appropriate conditions (e.g., hosts such as rice and corn and adequate amount of nitrogen), FA is modified into fusarinolic acid (FnA) and 9,10-dehydro-FA (Brown et al., 2012; Niehaus et al., 2014; López-Díaz et al., 2018).
The toxicity of FA, has been confirmed in plants, moulds, bacteria and in animals. FA disrupts the function of the cardiovascular, immune and nervous systems of animals. However, the interesting property of FA is its possible therapeutic effect for lowering blood pressure and potential anticarcinogenic activity in humans. In respect to its scientifically proven herbicidal, insecticidal, antifungal and antiviral activity and effective phytotoxicity against certain weeds, it has been used for the bio-control of weeds and plant protection (Wang and Ng, 1999; Song and Yee, 2001).
Moniliformin (MON, semisquaric acid, hydroxycyclobutenedione, C4H2O3) is a metabolite with a molecular weight of 98.1 g/mol that is soluble in water and polar solvents. It was first described as a phytotoxin because it inhibits the growth of a considerable number of plants. It is produced by more than 30 Fusarium species including F. acuminatum, F avenaceum, F. equiseti, F. moniliforme, F. oxysporum, F. proliferatum, F. solani and F. tricinctum. It has been detected in samples of corn and often in combination with FUM (Chelkowski et al., 1990; Jestoi, 2008; PubChem, 2025).
Data on biotransformation of this toxin are scarce, while toxicity is described in poultry (when feed was contaminated with 20 mg/kg). In the presence of other mycotoxins, toxic effects were more pronounced. It causes disruption of the heart function and blood circulation, followed by sudden death. Additionally, a common sign of intoxication is stunted growth and in the case of death, pathological changes in the heart and ascites can be found. Apart from birds (chickens, turkeys, ducks), heart failure has also been described in rats and is also associated with the development of cardiomyopathy and Kashin-Beck disease in humans (Leeson et al., 1995; Streit et al., 2013).
Other Fusarium mycotoxins
Of the 700 detected Fusarium metabolites, a large number are not comprehensively studied so there are no data on their occurrence, biotransformation and/or toxicity (Sulyok et al., 2020, 2024). In the last 15 years, the term “emerging mycotoxins” (EM) has been introduced in the scientific literature. The term occurred in the time when application of modern methods of mass spectrometry enabled simultaneous detection of a large number of moulds metabolites showing their common presence in food and feed. Likewise, new data confirmed that they are widespread and often in combination with well-studied mycotoxins of distinct toxic properties. Fusarium species also produce EM and these are the above described NIV, DAS, MON and FA, as well as apicidin, aureofusarin, beauvericin, butenolide, culmorin, 15-hydroxy-culmorin, enniatins (A, A1, B, B1), equsetin and fusarin C (IARC, 1993; Sulyok et al., 2006; Gallo et al., 2013; Streit et al., 2013; Kovalsky et al., 2016; Khoshal et al., 2019; EFSA, 2022; Siri-Anusornsak et al., 2022; Kolawole et al., 2024).
Among the Fusarium species, confirmed producers of EM toxins are F. acuminatum, F. avenaceum, F. cerealis, F. crookwellense, F. culmorum, F. equiseti, F, fujikori, F. graminearum, F. langsethiae, F. moniliforme, F. nivale, F. pallidoreseum, F. poae, F. proliferatum, F. roseum, F. sambucinum, F. semitectum, F. solani, F. subglutinans and F. tricinctum. Impacts on poultry health were recorded in studies on aureofusarin, beauvericin and butenolide. In swine, harmful chronic effects were described for culmorin, 15-hydroxy-culmorin, beauvericin, enniatins and their combinations with the previously described DON. In laboratory animals, toxic effects of EMs have been recorded mainly using high experimental concentrations. Additionally, apicidin, beauvericin, enniatins, equisetin and fusaric C may have antimicrobial effects on certain bacteria and moulds, while Fusarin C is also a potential carcinogen. Indeed, the concentrations of these toxins in food are generally low, significantly lower than those that cause effects in experimental (laboratory) animals. Hence, it is assumed that the toxic potential of EM and their combinations in animal feed is only likely after a long-term consumption of contaminated feed (IARC, 1993; Streit et al., 2013; Kolawole et al., 2024).
Occurrence of Fusarium mycotoxins
To present the occurrence of described mycotoxins, the data from global surveys confirm their continuous presence. For example, in a global survey that included analysis of 74,821 samples of feed and feed raw materials (maize, wheat, soybean) in the period from 2008 to 2017, at least one mycotoxin was detected in 88% of analysed samples (Gruber-Dorninger et al., 2019). In this survey, T-2 toxin was detected in the range from 2.0–48.2%, with median values of positive samples in the range 3.0–34.0 µg/kg. For DON, the prevalence was in the range from 23.1–84.8%, with median values of positive samples in the range from 28.0–505 µg/kg. FUM was detected in 22.2–81.8% of tested samples, with median values of positive samples in the range from 13–1,390 µg/kg. ZEN positive samples were in the range from 19.6–58.2%, with median values of positive samples in the range 1.5–105.0 µg/kg. In a more recent study on the global occurrence of mycotoxins in 6701 samples of analysed finished feed, wheat grains, maize kernels, and soybean in 2024, 25 of the most frequently found mycotoxins were reported, including their masked and modified forms and emerging mycotoxins. T-2 and HT-2 toxin were present in 10% and 14 % of samples with average concentrations of 24 µg/kg and 84 µg/kg, respectively. DON, 15-Ac-DON, DON-3-glucoside and NIV were detected in 78%, 16%, 31% and 10%, with average concentrations of 496 µg/kg, 192 µg/kg, 111 µg/kg and 198 µg/kg, respectively. FB1, FB2, FB3 and ZEN were present in 61%, 58%, 39% and 57%, with average concentrations of 448 µg/kg, 168 µg/kg, 97 µg/kg and 77 µg/kg, respectively. In the same study, BEA, enniatins (A, A1, B, B1) and MON were detected in the range from 63–70%, with an average concentration from 7–112 µg/kg (DSM-FIRMENICH, 2025). DAS and their metabolites were found in various cereals, baby food, beer, coffee, pulses, spices, feed and other food, ranging from 0 to 100% of 12,968 analysed samples, in a concentration range from 10–1,294.0 µg/kg (JECFA, 2017). NEO and FA are not routinely tested and their occurrence has been reported in 2.1% and 78.1% of analysed grain samples, at a concentration range from 5.7–23.9 µg/kg and from 31.4–205.0 µg/kg, respectively (EFSA, 2018a; Weaver et al., 2021).
From the presented overview of Fusarium species and their metabolites, the enormous progress in this field of research is evident. The use of modern scientific methodology has resulted in identification of diverse Fusarium species, toxigenic strains and detection of a large number of different metabolites. Unfortunately, most are still understudied. Further research of chemical characteristics, biosynthesis and modification processes, as well as their potential toxic properties, will elucidate the role of these substances in the life cycle of plants, and their significance in humans and animals. The fact that the occurrence of moulds and mycotoxins is inevitable, along with discoveries in this field of research, highlight the importance of further studies. Detection of newly discovered (toxigenic) Fusarium species and bioactive metabolites have advanced the understanding of mould pathogenicity and properties of metabolites, and provided new knowledge and uses of effective preventive measures that will ultimately reduce the anticipated detrimental effects in plants, animals and humans. Finally, it will also minimise economic losses in agriculture, animal husbandry and healthcare expenditures.
Conclusions
The presented description of Fusarium moulds, mycotoxins and metabolites has turned the spotlight on the need for precise identification of isolated moulds, using both morphological and molecular methods. Due to the confirmed correlation between mould virulence and their toxigenic potential, and the toxicity of mycotoxins, accurate identification and characterisation of individual metabolites is of paramount importance for the protection of plant, animal and human health. Accordingly, control programs needs to include routine analysis of raw materials and compound foods in the food production chain, risk assessment and application of effective preventive measures in animal husbandry. Precise and timely diagnostics, together with successful prevention will ultimately minimise the onset of diseases and intoxications in humans.
Acknowledgement
This study was funded by the European Union NextGenerationEU and supported by the Ministry of Science and Education of the Republic of Croatia through the project No. NPOO 10 of Croatian Veterinary Institute entitled “Biological diversity of moulds and occurrence of Fusarium mycotoxins and their metabolites in the most important cereals grown in the Republic of Croatia (MOULD_FUSion)”.
References [… show]
Fuzarijske plijesni, mikotoksini i njihovi metaboliti
Marijana Sokolović1* (dopisni autor), m_sokolovic@veinst.hr, orcid.org/0000-0003-3373-7415; Ana Vulić2, vulic@veinst.hr, orcid.org/0000-0002-9379-7236; Nina Kudumija2, kudumija@veinst.hr, orcid.org/0000-0001-6144-6514; Tina Lešić2, lesic@veinst.hr, orcid.org/0000-0001-6773-9473; Jelka Pleadin2, pleadin@veinst.hr, orcid.org/0000-0002-0768-0462; Marija Berendika1, mberendika@veinst.hr, orcid.org/0000-0002-6717-6292; Manuela Zadravec3, zadravec@veinst.hr.
1, Laboratorij za analitiku stočne hrane, Centar za peradarstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
2 Laboratorij za analitičku kemiju, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
3 Laboratorij za mikrobiologiju hrane za životinje, Odjel za veterinarsko javno zdravstvo, Hrvatski veterinarski institut, 10000 Zagreb, HrvatskaPlijesni roda Fusarium prisutne su u različitim vodenim i zemljanim okolišima kao patogeni, endofiti i/ili saprofiti. Lako se prilagođavaju različitim okolišnim uvjetima, te su stoga rasprostranjene u cijelom svijetu. Ipak, njihovom rastu i razvoju više pogoduju područja sa vlažnom i toplom klimom. Većina vrsta fuzarija su patogeni za biljke, ali neke mogu uzrokovati bolesti u ljudi i životinja bilo kao direktni patogeni ili djelovanjem njihovih metabolita, mikotoksina. Dokazano je da uzrokuju akutne i kronične intoksikacije te remete funkciju različitih organskih sustava. U najznačajnije skupine fuzarijskih mikotoksina ubrajamo trikotecene, fumonizine i zearalenone. Primjenom modernih tehnologija, instrumenata i metoda otkriven je veliki broj fuzarijskih plijesni i metabolita. Međutim, tokiskogene funkcije opisane su za manji broj vrsta, a za većinu novootkrivenih metabolita nema podataka o njihovim strukturnim i kemijskim karakteristikama, načinu biosinteze kao ni dokaz o potencijalnim (ne)poželjnim bioaktivnim učincima. S obzirom na neizbježnu svakodnevnu prisutnost fuzarija i/ili njihovih metabolita te na njihove potencijalne štetne učinke, neophodna su daljnja istraživanja novootkrivenih vrsta i metabolita. Cilj ovog preglednog rada je dati prikaz fuzarijskih vrsta i njihovih metabolita s osvrtom na njihove karakteristike rasta i biosintezu metabolita.
Ključne riječi: Fusarium, plijesni, mikotoksini, metaboliti, toksičnost