Histomonas meleagridis infections in turkeys
V.Mašović, B. Ječmenica*, T. Zglavnik, Š. Naletilić, F. Krstulović, L. Jurinović, R. Beck
Viktor Mašović1, masovic@veinst.hr, orcid.org/0009-0008-0744-5084; Biljana Ječmenica1, *(corresponding author), jecmenica@veinst.hr, orcid.org/0000-0003-2882-6528; Tihomir Zglavnik2, tzglavnik@veinst.hr; Šimun Naletilić3, naletilic@veinst.hr, orcid.org/0009-0002-5805-9892; Fani Krstulović1, fkrstulovic@veinst.hr, orcid.org/0009-0008-6270-5520; Luka Jurinović1, jurinovic@veinst.hr, orcid.org/0000-0002-6216-3978; Relja Beck4, beck@veinst.hr, orcid.org/0000-0002-5304-2875.
1 Laboratory for Bacteriology, Poultry Centre, Croatian Veterinary Institute, 10000 Zagreb, Croatia
2 Laboratory for Virology and Pathology, Poultry Centre, Croatian Veterinary Institute, 10000 Zagreb, Croatia
3 Laboratory for Pathology, Department of Pathological Morphology, Croatian Veterinary Institute, 10000 Zagreb, Croatia
4 Laboratory for Parasitology, Department of Bacteriology and Parasitology, Croatian Veterinary Institute, 10000 Zagreb, Croatia
https://doi.org/10.46419/cvj.57.4.4
Abstract
This report describes an infection of Histomonas meleagridis in a turkey flock kept together with pheasants and geese on a farm. The owner observed a number of turkey deaths and two turkey carcasses were sent to the Pathology Laboratory of the Poultry Centre of the Croatian Veterinary Institute for parasitological diagnosis. Histomonosis was confirmed through histopathological and parasitological methods. In total, 150 of 500 turkeys in the flock perished. The owner was advised to enhance biosecurity measures on the farm.
Keywords: histomonosis, biosecurity, turkey
Introduction
Histomoniasis, or blackhead disease, is one of the most common and widespread protozoal diseases in poultry. It is caused by the protozoan parasite Histomonas meleagridis. Turkeys are most susceptible to the disease between 3 and 12 weeks of age, while chickens are most susceptible at 4 to 6 weeks. Additionally, cases have been reported in other birds such as pheasants, peacocks, guinea fowl, and partridges (BIĐIN, 2008; DOLKA et al., 2015; BEER et al., 2022a). The disease was treatable until 2003, when the European Union banned all drugs against H. meleagridis for use in food-producing animals (HAUCK and HAFEZ, 2013). In layer and broiler breeder pullets, histomonosis can progress without obvious clinical signs and with low mortality; however, in young animals, cases have been reported with high mortality (MCDOUGALD, 2005). The disease has two characteristic morphological forms: an ameboid and a flagellated form. Transmission of H. meleagridis occurs in two ways. First, the parasite can spread via a vector, the coeliac nematode Heterakis gallinarum. This transmission requires consumption of histomonds by H. gallinarum and their uptake into its ova. H. gallinarum creates a safe environment for histomonads to survive outside the host. Birds can become infected by ingesting H. gallinarum eggs, and some species such as chickens act as mechanical vectors, spreading infected eggs. Second, H. meleagridis can spread laterally without H. gallinarum, through drinking from the cloaca within a flock. Flies, earthworms, and other invertebrates contribute to the spread of infected H. gallinarum eggs (MCDOUGALD, 2005; BEER et al., 2022a). The first signs of the disease in turkeys appear nine days after infection, including sulfur-coloured diarrhea, lethargy, anorexia, and depression. The first deaths occur after 12 days, with a peak between 19 and 21 days after infection (HAUCK and HAFEZ, 2013; EL-WAHAB et al., 2021).
Material and methods
Case description
The farm in Koprivničko-Križevačka County houses a total of 1000 pheasants and 100 geese, separated only by wire. They are housed in aviaries and fed commercial feed. Drinkers and feeders are placed on the floor within the aviaries. In May, the owner decided to add 500 three-month-old turkeys to the farm. Fourteen days after purchase, some 20 turkey deaths were observed. Over the next two days, the death toll rose by 40 animals. An outbreak of a rapidly progressing disease resulted in a total of 150 dead turkeys, causing significant economic losses. The owner began treatment with the antibiotic Tylosin at his own initiative, but the turkey deaths continued, prompting him to seek professional help. Samples of liver and cecum were taken and fixed in 10% buffered formalin for 24 hours. Standard histological protocol was followed, including dehydration, embedding in paraffin blocks, and sectioning into serial 4 µm thick slices. Sections were stained with hematoxylin and eosin (H&E) and examined under a microscope (Zeiss Axio Imager.A2). Microscope images were captured using a Digicyte BigEye camera.
Necropsy diagnosis
Turkey carcass 1
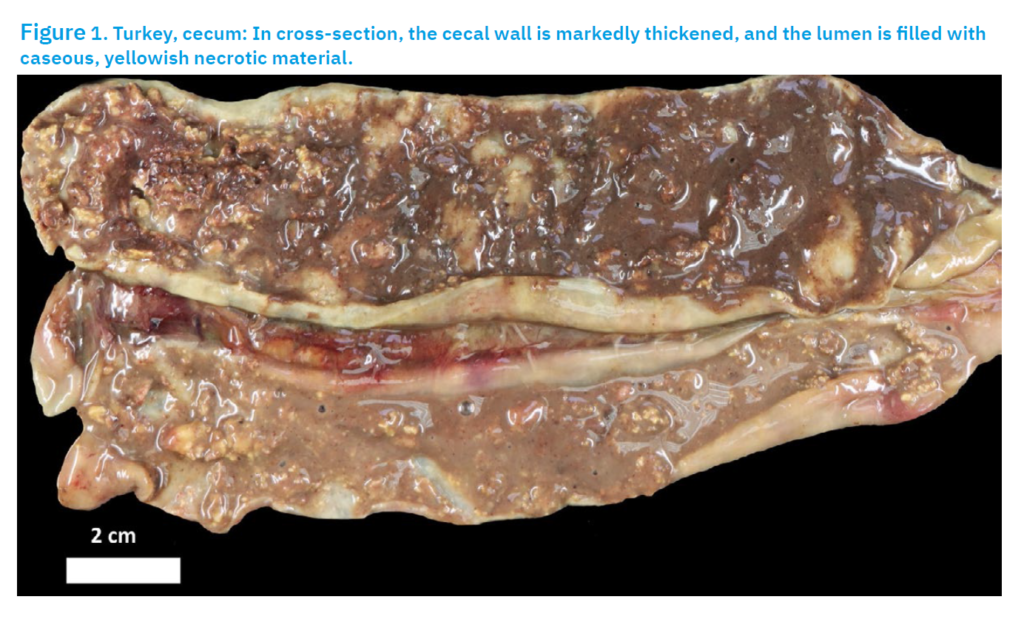
Carcass 1 was in moderate cachexia. The intestines were enlarged and had cheesy to fibro necrotic cores in the lumen, which had become inflamed. The liver was enlarged and green to bronze in colour. There were numerous round, sunken, golden yellow or pale necrotic foci in the liver, reminiscent of a volcanic crater. The necrotic foci were up to one centimetre in size. TURKEY CARCASS 2Carcass 2 showed moderate cachexia. Haemorrhages were noted throughout the inner thigh muscles. The liver was enlarged and had numerous ulcerated lesions on the surface of the liver. The lesions were green and crater-like in shape. The crop contained a green-yellow liquid with an intense sour odour. The intestines and caecum were inflamed. There were also cheesy, cheese-like cores present in the intestines (Fig. 1). The caecal content had a foul-smelling odour.

Patohistology
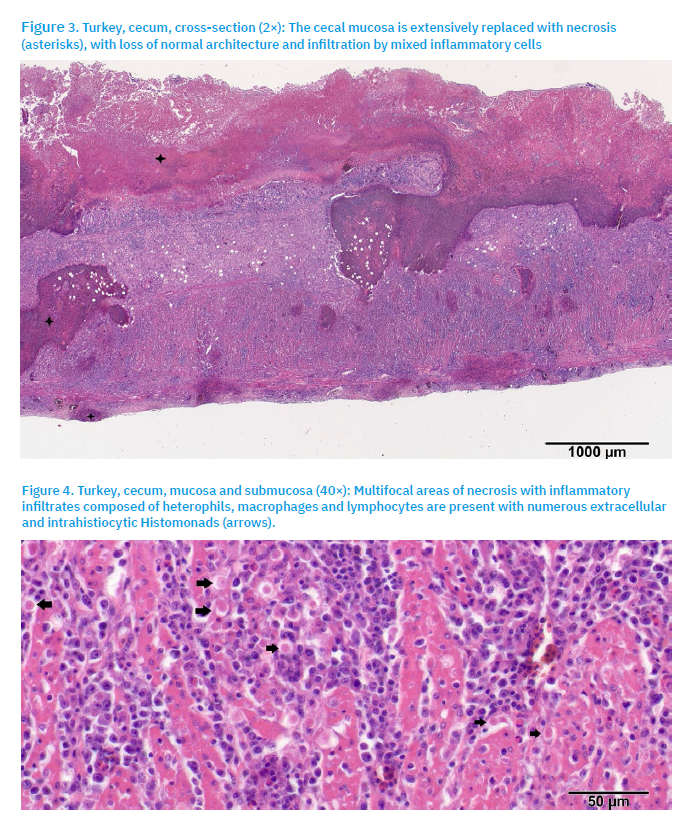
Diffuse areas of lytic necrosis affected the cecal mucosa, extending into the submucosa (Fig. 3). The necrotic mucosa appeared eosinophilic and amorphous, with scattered basophilic nuclear fragments. The necrotic regions, along with the submucosa and muscularis, were infiltrated by a mixed population of inflammatory cells, including heterophils, histocytes, lymphocytes, and occasional plasma cells. Mild haemorrhaging and oedema were also present in the surrounding connective tissue. Numerous protozoal trophozoites (histomonads) were observed both extracellularly and intrahistiocytically (Fig. 4), and were irregularly shaped, round to oval, 10-20 µm in diameter, with bright eosinophilic cytoplasm and a centrally located dark basophilic nucleus.
Parasitology diagnosis
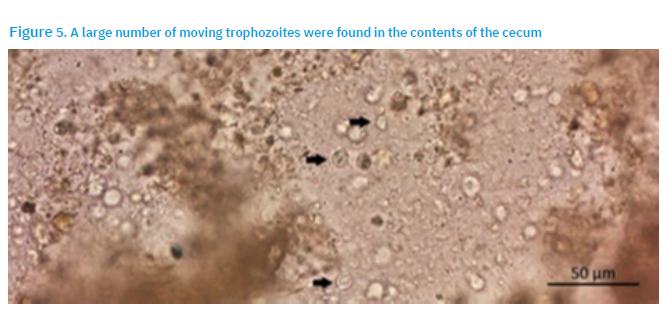
Surface imprints were prepared from the liver lesions, air-dried and stained using May-Grünwald-Giemsa. Mucosal scrapings from the caeca were placed on the slides, and covered with a cover slide. Morphology was examined under an Imager M.2 microscope (Zeiss) equipped with Axiovision and ZEN2 Pro software. A large quantity of trophozoites were found by microscopic examination (Fig 5).
Discussion
In the studied histomonosis outbreak, about 150 turkeys perished, leading to significant economic losses. Unfortunately, the farmer treated the animals himself but sought professional help only after mass mortality began, which allowed the disease to spread rapidly among the animals. It is likely that the turkeys were infected on the farm, and the pathogens were not introduced with the new turkeys. This is supported by the fact that histomonosis was also found in pheasants on the farm, which are more resistant and less likely to become ill, suggesting they were likely the source of the infection (BIĐIN, 2008; BEER et al., 2022a). Additionally, about three weeks passed since the arrival of the turkeys at the farm, sufficient time for H. meleagradis to multiply and spread through direct contact within the flock. There is a possibility of co-infection with H. meleagridis and Tetratrichomonas gallinarum, based on characteristic macroscopic liver changes and parasitological diagnosis, but no further diagnostics such as Polymerase Chain Reaction (PCR) were performed in this case. According to SHCHEBENTOVSKA and HOLUBTSOVA (2020), infection with H. meleagridis causes an enlarged appendix with diffuse hemorrhages and nodules. Histomonosis also results in an enlarged liver and caseous, cheese-like caecal cores, which were present here. Although T. gallinarum is not considered highly pathogenic, fatal cases have been reported in black swans (Cygnus atratus) (FENG et al., 2021), wild purple finches (Carpodacus purpureus), and American goldfinches (Carduelis tristis) (FORZÁN et al., 2010). This outbreak occurred in summer 2024, and later in autumn, another case of H. meleagridis was diagnosed in a pheasant carcass during post-mortem examination, which showed characteristic gross lesions in the liver. No further details about pheasant deaths are available. BIĐIN (2008) discusses differences in clinical symptoms across bird species, noting that pheasants exhibit milder signs compared to turkeys, as seen in this case. Previous vaccination studies have not shown effective prevention of histomonosis, though recent attempts using attenuated H. meleagridis have yielded some protection in turkeys and chickens. Nonetheless, the immune response against histomonads remains unresolved (BEER et al., 2022b). Currently, the most effective measures against histomonosis include farm management practices such as thorough cleaning and disinfection, raising different poultry species and age groups separately, installing barriers within the housing, and changing litter after an outbreak, though these are not always completely effective (REGMI et al., 2016). The farmer was advised to remove all drinkers and feeders for two weeks and clean the barn with a 10% chlorine solution. Animals displaying clinical signs should be housed separately to prevent further spread. Additionally, the flock should be supplemented with a vitamin complex containing AD3E and vitamin C to boost overall resistance. Since the main transmission route involves H. gallinarum, regular deworming is necessary.
Conclusion
This report documents an infection with H. meleagridis in turkeys. It clearly highlights the importance of strict biosecurity measures, as there are no available drugs or vaccines to prevent or treat histomonosis in turkeys. Consequently, the economic impact can be severe, as this case demonstrates.
References [… show]
Infekcija s Histomonas meleagridis u purana – prikaz slučaja
Viktor Mašović1, masovic@veinst.hr, orcid.org/0009-0008-0744-5084; Biljana Ječmenica1, *(dopisni autor), jecmenica@veinst.hr, orcid.org/0000-0003-2882-6528; Tihomir Zglavnik2, tzglavnik@veinst.hr; Šimun Naletilić3, naletilic@veinst.hr, orcid.org/0009-0002-5805-9892; Fani Krstulović1, fkrstulovic@veinst.hr, orcid.org/0009-0008-6270-5520; Luka Jurinović1, jurinovic@veinst.hr, orcid.org/0000-0002-6216-3978; Relja Beck4, beck@veinst.hr, orcid.org/0000-0002-5304-2875.
1 Laboratorij za bakteriologiju, Centar za peradarstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
2 Laboratorij za virologiju i patologiju, Centar za peradarstvo, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
3 Laboratorij za patologiju, Odjel za patološku morfologiju, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
4 Laboratorij za parazitologiju, Odjel za bakteriologiju i parazitologiju, Hrvatski veterinarski institut, 10000 Zagreb, HrvatskaOvaj rad prikazuje slučaj infekcije s Histomonas meleagridis u jednom jatu purana. Jato purana potječe s farme gdje je zajedno držano s fazanima i guskama. Nakon nekoliko uginuća jedinki, vlasnik traži stručnu pomoć te dvije lešine šalje na Hrvatski veterinarski institut – podružnica Centar za peradarstvo. Nakon obavljene parazitološke i histopatološke pretrage, dijagnosticirana je histomonijaza. Ukupno, vlasniku je uginulo 150 od 500 purana iz istoga jata. Stručna pomoć mu je pružena u vidu savjete oko poboljšavanja biosigurnosnih mjera na farmi i poboljšanja tehnoloških uvjeta proizvodnje životinja.
Ključne riječi: histomonijaza, biosigurnost, puran