Prevalence and antimicrobial susceptibility profiles of Staphylococcus spp. isolated from broilers in Croatia
T. Amšel Zelenika, M. Balenović*, M. Berendika, F. Krstulović, M. Kraljević, and M. Sokolović
Tajana Amšel Zelenika1, t_amsel-zelenika@veinst.hr, orcid.org/0000-0002-7038-2433; Mirta Balenović1*, (corresponding author), m_balenovic@veinst.hr, orcid.org/0000-0002-3966-1389); Marija Berendika1, mberendika@veinst.hr, orcid.org/0000-0002-6717-6292; Fani Krstulović1, fkrstulovic@veinst.hr; orcid.org/0009-0008-6270-5520; Mate Kraljević2, mate.kraljevic@praviritam.hr; Marijana Sokolović1, m_sokolovic@veinst.hr, 0000-0003-3373-7415.
1Croatian Veterinary institute, Poultry Centre Zagreb, Croatia;
2Pravi ritam Ltd, Zagreb, Croatia
https://doi.org/10.46419/cvj.57.4.8
Abstract
Staphylococcal infections can be a serious public health problem for farm workers and other persons in contact with poultry, due to their ability to cause a wide range of infections in other animal species and humans. In addition, the presence of pathogenic bacteria in the intestines of poultry can lead to contamination of poultry products. Coagulase-positive (CoPS) and coagulase-negative (CoNS) staphylococci, which are potentially involved in staphylococcal infections in humans, have been associated with poultry. The best-known CoPS is Staphylococcus aureus (S. aureus). The aim of this study was to evaluate the prevalence of Staphylococcus spp. in broilers on commercial farms in Croatia, and to determine their antimicrobial susceptibility profiles. Staphylococcus spp. strains isolated from cloacal swabs of apparently healthy broiler flocks in Croatia were analysed, and a relatively high Staphylococcus infection rate was detected, with a total percentage of isolated Staphylococcus strains of 38%. Among these, 3/100 (3%), 5/100 (5%), 3/100 (3%), 2/100 (2%) were S. aureus, and three CoNS: S. sciuri, S. simiae and S. lentus, respectively. The majority of isolated strains were resistant to penicillin (88.8%), clindamycin (85%), cefoxitin (78%), trimethoprim/sulfamethoxazole (65%), ciprofloxacin (53%), erythromycin (40%) and tetracycline (25%). The highest susceptibility of isolated strains was to chloramphenicol (95%) and gentamicin (89%). All S. aureus, S. lentus and S. simiae isolates were multi-drug-resistant strains that exhibited resistance to three or more antimicrobials, while S. sciuri isolates were resistant to two of the tested antimicrobials.
Keywords: Staphylococcus, broiler, antimicrobial resistance, S. aureus, S. simiae, S. sciuri, S. lentus
Introduction
Staphylococcus species are Gram-positive bacteria with a broad host spectrum including humans and birds, colonizing skin, mucosa and respiratory tract (Geenen et al., 2013; Ribeiro et al., 2018). Although primarily considered commensal organisms, certain strains are pathogens, especially in the case of a host with a compromised immune system (Gibson et al., 2021). Currently, the genus Staphylococcus includes 92 recognized species and 30 subspecies (LPSN, retrieved on 2 September 2025). Staphylococci are common inhabitants of the intestinal tract of poultry (Salanitro et al., 1974; Devriese et al., 1975; Salanitro et al., 1978), which can pose a serious public health problem, especially for farm workers, but also for all people coming into close contact with animals (Zaheer et al., 2017). Infection can be transmitted through skin contact, especially via damaged skin (cuts, scratches, ulcers, etc.) (Varela et al., 2022). Furthermore, the presence of bacteria in the poultry intestines increases the risk of contamination of poultry products (Pan and Yu, 2014). Contaminated poultry products are a potential source of infection with resistant strains for other animals or humans. In addition, their ability to produce enterotoxins can result in foodborne intoxications in humans (Khairulla et al., 2024). Improper processing of raw meat can lead to cross-contamination, and consumption of inadequately thermally processed meat can lead to infection or poisoning in humans (Kluytmans, 2010; Sergelidis et al., 2017). This risk is more significant in the case of multidrug resistant bacteria. The widespread use of antimicrobials in animals, including poultry, has contributed to the ability of Staphylococcus spp. to acquire a variety of resistance genes (Otalu et al., 2011). Staphylococcal infections in animals and humans are commonly treated with antimicrobials, usually β-lactam antibiotics. These antibiotics were originally very effective against staphylococci, but in the mid-1940s, β-lactamase-producing Staphylococcus isolates emerged and their prevalence increased dramatically within a few years (Liu et al., 2012; Geenen et al., 2013). Staphylococcus spp., particularly strains of S. aureus, are known to produce β-lactamases and to acquire and spread various types of resistance genes via mobile genetic elements, plasmids and transposons, which play an important role in the emergence of multidrug resistance (McCallum et al., 2010).
From a clinical perspective, harmful species include coagulase-positive staphylococci (CoPS) such as S. aureus, S. intermedius, S. schleiferi subsp. coagulans, S. pseudointermedius, S. lutrae, S. delphini and some strains of S. hyicus (Pottumarthy et al., 2004; Van Hoovels et al., 2006). As an opportunistic pathogen in humans and animals, S. aureus is mainly associated with food poisoning, as it can produce up to 25 different toxins (Ortega et al., 2010; Aydin et al., 2011), but it can also cause a number of infections and diseases such as dermatitis, pneumonia, meningitis, osteomyelitis in humans and bumblebee disease in poultry (Quinn et al., 2000). One of the most important and best-known pathogens worldwide is methicillin-resistant S. aureus (MRSA). It has been detected in various animal species, poultry and humans, but also in animal products, especially poultry. Humans become infected with MRSA strains through improper handling of infected animals or poultry carcasses. Furthermore, live poultry can also transmit MRSA via close contact with poultry farm employees.
Coagulase-negative staphylococci (CoNS) can cause hospital-acquired infections, but they also occur as a rare cause of food poisoning (Tong et al., 2015). Various studies on CoNS have shown the increasing medical importance of this Staphylococcus subtype in humans (Becker et al., 2014). CoNS have fewer virulence factors involved in the pathogenesis of infection compared to S. aureus, but have recently become a common cause of nosocomial infections. In addition, increasing rates of antibiotic resistance have been observed in CoNS, in some cases even higher than in S. aureus, limiting the available therapeutic options (Heilmann et al., 2019). The most important virulence factor of this group of bacteria is their ability to form complex biofilm structures on the surface of damaged tissue (de Silva et al., 2002).
One of the more interesting CoNS is S. simiae, which so far has only been described as an isolate from the gastrointestinal tract of South American squirrel monkeys (Saimiri sciureus L.) (Pantůček et al., 2005). To the extent of our knowledge and the available scientific literature, there are no studies on the significance of S. simae in poultry. Furthermore, S. simiae has not been established as a cause of infections in humans, and the majority of clinical staphylococcal infections in humans are related to other species, primarily S. aureus and S. pseudintermedius (Van Hoovels et al., 2006). A genome-based phylogeny of the genus has confirmed that S. simiae is a sister group to S. aureus, indicating that the most basal Staphylococcus lineage is S. pseudintermedius, followed by S. carnosus (Suzuki et al., 2012). This research has proven that genes associated with pathogenesis, especially virulence factors, are not the result of vertical inheritance alone, but are often imported into the genome through horizontal gene transfer, which proves that bacteria exchange genes between species and even between different lineages within the genus through mobile genetic elements (e.g. plasmids, transposons, phages). Therefore, horizontal gene transfer might be responsible for development of new traits such as antimicrobial resistance, increased virulence and the like, giving this species special significance.
- sciuri is commonly found on the skin and mucous membranes of warm-blooded animals and humans (Adegoke, 1986; Hauschild and Schwarz, 2003; Shittu et al., 2004; Huber et al., 2011) and in the environment (Pioch et al., 1988). Until recently, it was considered a non-pathogenic commensal bacterium, though it was associated with various diseases in animals. In humans, various infections have been described, including endocarditis (Hedin and Widerström, 1998), wound infections (Kolawole and Shittu, 1997), peritonitis (Wallet et al., 2000), septic shock (Horii et al., 2001) and urinary tract infections (Stepanovic et al., 2003).
- lentus is also occasionally found in poultry, but it is significant that S. lentus is a very common isolate among staphylococci in humans. Al-Taii et al. (2024) investigated the prevalence of staphylococci in birds and their breeders. In humans, S. lentus was most prevalent (42.85%), while in birds it was detected in 10% of analysed swabs. All isolates had a moderate ability to form biofilms. Isolates isolated from human swabs showed that S. lentus was resistant to seven antimicrobials tested (methicillin, oxacillin, gentamycin, trimethoprim/sulfamethoxazole, chloramphenicol, ciprofloxacin and doxycycline) in this study, while in poultry S. lentus isolates were resistant only to methicillin and oxacillin (20%) and to chloramphenicol (40%).
Given the lack of studies on the isolation and antimicrobial susceptibility of Staphylococcus spp. from the intestines of broiler chickens in Croatia, which are a potential source of infection for humans, and for contamination of poultry meat, we investigated the prevalence and antimicrobial resistance profiles of Staphylococcus spp. from cloacal swabs of seemingly healthy broiler flocks, with a focus on the species S. aureus, S. sciuri, S. lentus and S. simiae.
Materials and Methods
Sample collection, isolation, identification to the genus level, and determination of antimicrobial resistance profiles were performed in Poultry Centre, Croatian Veterinary Institute in Zagreb, Croatia. Confirmation of isolate identification to the species level was performed at the Ruđer Bošković Institute in Zagreb, Croatia.
Sample collection
A total of 100 cloacal swabs were sampled from five randomly selected broiler farms in central and eastern Croatia in the March 2025 (20 cloacal swabs per farm). All animals were in good health at the time of sampling. The samples were stored in a cool box with an ice pack, transported to the laboratory, and analysed immediately upon their collection.
Isolation and identification of bacteria
Isolation, identification and phenotypic susceptibility testing of MRSA and related species was performed according to the recommendations given by EFSA (2022) and EURL-AR (2024). In short, a pre-enrichment step was performed on individual single anal swab by adding 5 mL Mueller-Hinton broth (Thermo Fisher Scientific Inc., USA) containing 6.5% sodium chloride and incubating at 36 ± 1°C for 16–24 h, and additional incubation for subsequent 16–24 h. After incubation, 10 μL pre-enrichment culture was cultured on selective agar plates CHROMagar™ MRSA (CHROMagar, France) for isolation and differentiation of methicillin-resistant S. aureus (MRSA), and incubated at 36 ± 1°C for 16–24 h. Presumptive colonies were subcultured (one presumptive colony from each chromogenic plate) on Brilliance™ MRSA 2 Agar (Thermo Fisher Scientific Inc., USA), Baird-Parker agar (Thermo Fisher Scientific Inc., USA), Columbia Agar (Thermo Fisher Scientific Inc., USA), Columbia Blood Agar (Thermo Fisher Scientific Inc., USA), EKA agar (Thermo Fisher Scientific Inc., USA) and Mannitol Salt Agar (Thermo Fisher Scientific Inc., USA) for evaluation of their morphology and haemolysis.
Confirmation of isolated colonies was made by gram stains, catalase, oxidase and Staphylase™ (Thermo Fisher Scientific Inc., USA) tests. Species levels of isolates were confirmed using Matrix-Assisted Laser Desorption/Ionization Time-Of-Flight (MALDI-TOF) Mass spectrometry (Bruker Daltonics, Germany).
For further genotypic testing of MRSA isolates to be performed as continuation of this study, all isolated target strains were stored according to the guidelines described by European Union Reference Laboratory (EURL-AMR, 2025).
Antimicrobial Susceptibility testing was performed to evaluate whether isolates belong to livestock-associated strains and whether they are resistant to selected antimicrobials, including those that are considered as particularly important in epidemiology of human infections (tetracycline, linezolid and vancomycin). For that purpose, the EFSA (2022) and EURL-AR (2024) guidelines were followed. Tests were performed by the disk diffusion method in compliance with Performance Standards for Antimicrobial Disk Susceptibility Tests (CLSI-M02, 2024) and EUCAST disk diffusion method (EUCAST, 2025a) for a recommended panel of antimicrobials: chloramphenicol (BioMaxima, Poland), ciprofloxacin (Bioanalyse, Turkey), gentamicin (Bioanalyse, Turkey), clindamycin (BioMaxima, Poland), erythromycin (BioMaxima, Poland), cefoxitin (Thermo Fisher Scientific Inc., USA), oxacillin (Thermo Fisher Scientific Inc., USA), penicillin (Thermo Fisher Scientific Inc., USA), trimethoprim/sulfamethoxazole (Thermo Fisher Scientific Inc., USA) and tetracycline (BioMaxima, Poland). The results were evaluated according to Performance Standards for Antimicrobial Susceptibility Testing (CLSI-M100, 2025) and EUCAST (2025b).
Results
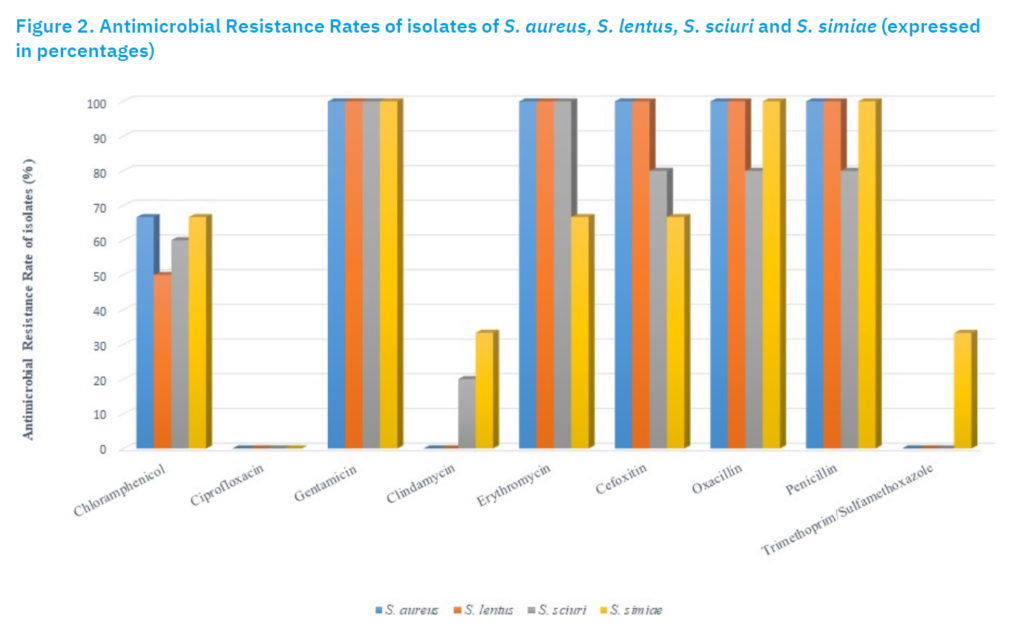
Results of isolation and identification of isolates Staphylococcus strains were isolated from 38% of samples. Of these, 3/38 (7.9%), 5/38 (13.2%), 3/38 (7.9%), 2/38 (5.3%) 25/38 (68.4%) were S. aureus, S. sciuri, S. simiae and S. lentus (Table 1). The identity of isolates was adequately confirmed to the genus level, including for the isolated S. aureus strains. Identification of other isolates to the species level based on morphology, biochemical tests and MALDI-TOF provided only presumptive results, since repeated testing of isolates using mass spectrometry did not result with high-confidence level of their identification (score ≥ 2.00).
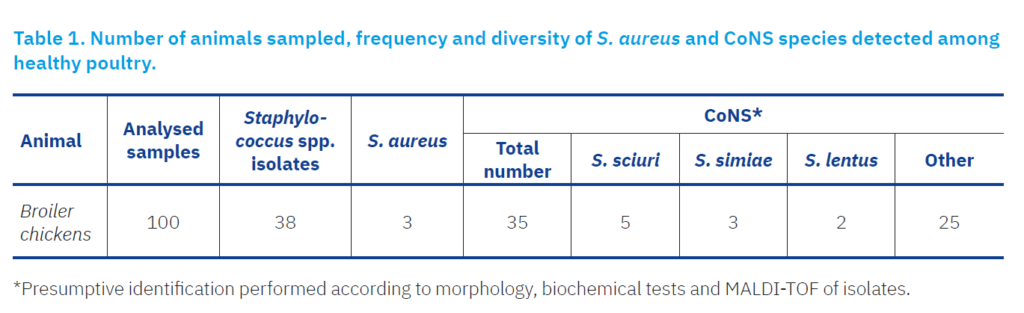
Results of determination of antimicrobial resistance profiles of isolates
Ten antimicrobials were tested, and the isolates showed highest resistance to penicillin (88.8%), followed by clindamycin (85%), oxacillin (80%), cefoxitin (78%), trimethoprim/ sulfamethoxazole (65%), ciprofloxacin (53%), erythromycin (40%) and tetracycline (25%). The isolates showed highest sensitivity to chloramphenicol (95%) and gentamicin (89%) (Figure 1).
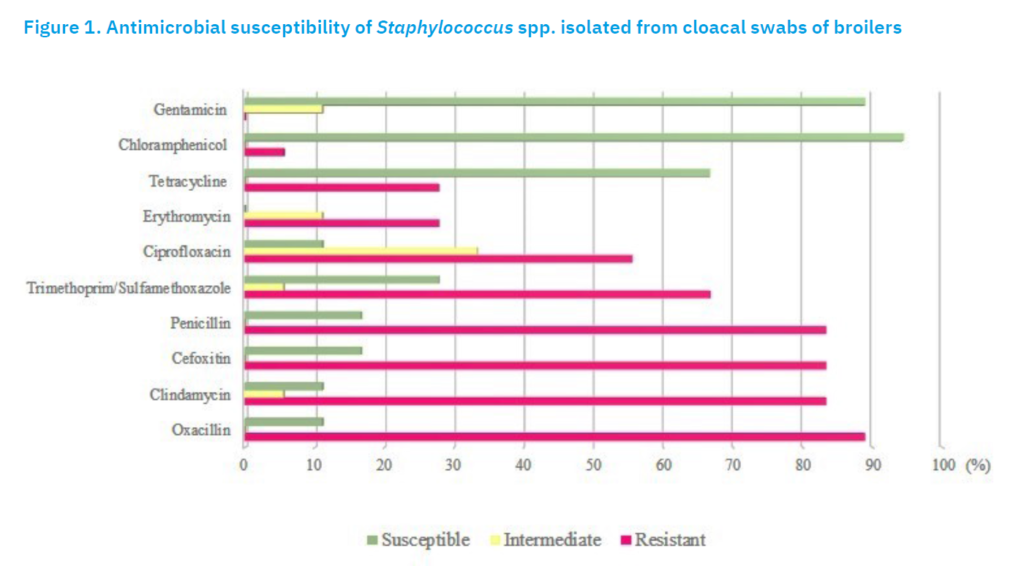
Discussion
A relatively high incidence (38%) of isolates of Staphylococcus spp. was found in broilers (Table 1). Saleh et al. (2003) reported that the prevalence of Staphylococcus isolates in broilers was 32%, which is in compliance with our results in broilers. Marek et al. (2016) found only 10.8% staphylococcal infections in poultry in Poland, though the percentage of isolates from broilers was 41.84% and therefore similar to that reported here. Pimenta et al. (2021) investigated the prevalence of staphylococci and their phenotypic resistance to β-lactam drugs in cloacal and tracheal swabs from 220 isolates of broiler chickens on two farms in the mountainous region of Rio de Janeiro, Brazil. A total of 88 strains of Staphylococcus spp. were isolated from 120 cloacal and tracheal samples. In the cloacal swabs of both broiler farms, 42.05% (37/88) of the Staphylococcus isolates were found, which is only 4% higher more than in our study. Syed et al. (2020) investigated the difference in the incidence of staphylococci from intestines of farmed and household chickens. A total of 100 poultry intestines (50 each from farmed and household chickens) were collected from poultry meat shops in Haripur and the Abbottabad districts of Khyber Pakhtunkhwa province of Pakistan where they were slaughtered. Their result was much higher than reported here, with a prevalence of 70% in commercial poultry (35/50) and 64% (32/50) in extensively reared poultry. Sukalić et al. (2024) found only a 1.5% prevalence of Staphylococcus spp. in 68 dead broiler chickens. Among the staphylococci in this study, 7.9% strains were S. aureus indicating 3% prevalence in cloacal swabs from broiler chickens (Table 1). In antimicrobial susceptibility testing, all S. aureus isolates were resistant to clindamycin, cefoxitin, oxacillin, penicillin and trimethoprim/sulfamethoxazole, and susceptible to chloramphenicol, gentamicin, erythromycin and tetracycline (Figure 2). Salama et al. (2021) investigated the prevalence of S. aureus in poultry in southern Egypt. Their results, based on cultural and biochemical characteristics, showed that about 10% (40/405) of all samples from poultry were identified as S. aureus. Most samples were isolated from chickens such as broilers and cockerels with 12% (38/315) of the total prevalence, followed by geese with 5.6% (2/36) and samples from manual slaughterhouses with 12.5% (2/124). In contrast to our study, Salama et al. (2021) reported susceptibility to trimethoprim/sulfamethoxazole but high resistance to erythromycin and tetracycline.
It has been described in the literature that S. lentus, as a CoNS, has the ability to form complex biofilm structures on the surface of damaged tissue (de Silva et al., 2002), making this its main virulence factor. Marek et al. (2016) reported a similar prevalence of S. lentus in broilers (5%) as reported here (2%) (Table 1). However, the proportion of S. lentus among the positive isolates was 5.3%. Al-Taii et al. (2024) demonstrated in a study conducted on birds and their breeders that isolated strains of S. lentus from poultry showed 20% resistance to methicillin and oxacillin and 40% to chloramphenicol. In the present study, all S. lentus isolates were resistant to clindamycin, cefoxitin, oxacillin, penicillin, trimethoprim/sulfamethoxazole, intermediate to ciprofloxacin, and susceptibile to chloramphenicol, gentamicin, erythromycin and tetracycline (Figure 2). In our study, among Staphylococcus isolates, presumptive identification at the species level showed a 13.2% incidence of S. sciuri (Table 1). Nemeghaire et al. (2014) investigated the prevalence of methicillin-resistant S. sciuri strains in poultry, as these can represent a reservoir of resistance genes for other strains. Of the 200 nasal swabs analysed, taken from Belgian broiler farms, 25 isolates (12.5%) of methicillin-resistant S. sciuri were detected. All these isolates were resistant to fusidic acid, tiamulin and gentamicin and susceptible to rifampicin and vancomycin. As shown in Figure 2, all our S. sciuri isolates were resistant to clindamycin and cefoxitin, highly resistant to oxacillin, trimethoprim/sulfamethoxazole and penicillin and susceptible to gentamicin, tetracycline and chloramphenicol. According to the currently available data, there are no studies on the incidence and antimicrobial resistance of S. simiae isolates in poultry or the isolation of S. simiae from cloacal swabs of broiler chickens. In the present study, among the isolated staphylococci from cloacal swabs from broiler chickens, 7.9% were confirmed as S. simiae (Table 1). Results of antimicrobial susceptibility testing revealed that all strains were resistant to clindamycin, penicillin, trimethoprim/sulfamethoxazole and the majority were resistant to ciprofloxacin, cefoxitin and oxacillin (Figure 2). Furthermore, all S. simiae isolates were susceptible to chloramphenicol and gentamicin. According to the current knowledge, S. simiae is not considered an important potential pathogen for poultry and humans. According to Suzuki et al. (2012), pathogenesis in the S. aureus group evolved after the divergence of S. simiae and S. aureus from their common ancestor by the horizontal transfer of mobile genetic elements. Among the suggested CoNS isolates, this study has confirmed the incidence of S. lentus, S. sciuri and S. simiae. While S. lentus and S. sciuri have been reported in other studies, it is also possible that our isolates belong to these two species. In the case of S. simiae, the result is less likely since this species has not been confirmed in samples of similar origin. However, all isolated S. simiae strains were diverse in respect to their morphology, mass-spectrometry results and antimicrobial resistance profiles, indicating that even if they cannot be designated as S. simiae, they are different from the isolated S. aureus. Further genetic studies are necessary to uniquely identify these isolates and to provide information regarding their importance for humans and animals.
Observed differences in our results regarding the incidence of staphylococci and their antimicrobial resistance profile in comparison with the results of other research groups might be explained by the different testing methods and protocols, and the growing trend of emerging resistance in bacteria. Furthermore, our results confirm the statements of Salanitro et al. (1978) that the gut of healthy chickens is filled with a complex microbiota and that the types of microorganisms present in the gut of poultry depend largely on the diet, environment and surroundings in which they live. The role of poultry as a potential source of infection by antibiotic-resistant Staphylococcus strains in humans is certain and merits further study. The high rate of resistance to the tested antimicrobials, as reported here, indicates a serious health threat to both animals and humans. Future research should include data on the use of antimicrobials for the treatment of staphylococcal infections in poultry and humans, to improve knowledge on resistance dynamics to individual antimicrobials. Finally, it is clear that the use of antimicrobials requires stricter controls to prevent further development of bacterial resistance. In addition, our results highlight the increasing role of CoNS infections in poultry, especially since the majority were multi-drug-resistant isolates (with resistance to five or more antimicrobials). The importance of these findings is in the numerous studies on CoNS nosocomial infections in humans having a high mortality rate (de Silva et al., 2002; Piette and Verschraegen, 2009). Since the genes responsible for the trait of antimicrobial resistance can be spread by mobile genetic elements, plasmids and transposons (McCallum et al., 2010), the presence of potential pathogenic staphylococci in poultry may have a contributing role in the emergence of multiple antimicrobial resistance in other bacteria using various mechanisms (including gene transfer through transduction, conjugation or transformation).
References [… show]
Prevalencija i antimikrobna osjetljivost Staphylococcus spp. u tovnih pilića u Hrvatskoj
Tajana AMŠEL ZELENIKA1, t_amsel-zelenika@veinst.hr, orcid.org/0000-0002-7038-2433; Mirta BALENOVIĆ1*, (dopisni autor), m_balenovic@veinst.hr, orcid.org/0000-0002-3966-1389); Marija BERENDIKA1, mberendika@veinst.hr, orcid.org/0000-0002-6717-6292; Fani KRSTULOVIĆ1, fkrstulovic@veinst.hr; orcid.org/0009-0008-6270-5520; Mate KRALJEVIĆ2, mate.kraljevic@praviritam.hr; Marijana SOKOLOVIĆ1, m_sokolovic@veinst.hr, 0000-0003-3373-7415.
1Centar za peradarstvo, Hrvatski veterinarski institut, Zagreb, Hrvatska
2Pravi ritam d.o.o., Zagreb, HrvatskaStafilokokne infekcije predstavljaju ozbiljan javnozdravstveni problem za osobe koje se bave uzgojem životinja kao i ostale koje dolaze u bliski kontakt s peradi budući da ove bakterije mogu izazvati širok raspon infekcija i u drugih vrsta životinja i ljudi. Osim toga, prisutnost patogenih bakterija u crijevima peradi može dovesti do kontaminacije peradarskih proizvoda. Koagulaza-pozitivni (CoPS) i koagulaza-negativni (CoNS) stafilokoki, koji su mogući uzročnici stafilokoknih infekcija u ljudi, mogu uzrokovati sojevi koji uzrokuju bolest u peradi. Najpoznatiji CoPS je Staphylococcus aureus (S. aureus). Cilj ovog istraživanja bio je procijeniti prevalenciju Staphylococcus spp. u zdravih brojlera na komercijalnim farmama u Hrvatskoj i utvrditi njihovu osjetljivost na antimikrobne pripravke. Pritom smo analizirali izolate iz kloakalnih briseva naizgled zdravih jata tovnih pilića u Hrvatskoj. Rezultati našeg istraživanja pokazali su relativno visoku stopu infekcije stafilokokom kod tovnih pilića. Ukupni postotak izoliranih sojeva stafilokoka bio je 38,0%. Od njih, 3/100 (3%), 5/100 (5%), 3/100 (3%), 2/100 (2%) bili su S. aureus, te CoNS: S. sciuri, S. simiae i S. lentus. Većina izoliranih sojeva bila je otporna na penicilin (88,8%), klindamicin (85%), cefoksitin (78%), trimetoprim/sulfametoksazol (65%), ciprofloksacin (53%), eritromicin (40%) i tetraciklin (25%). Najveća osjetljivost izoliranih sojeva bila je na kloramfenikol (95%) i gentamicin (89%). Svi izolati S. aureus, S. lentus i S. simiae bili su sojevi otporni na više antimikrobnih pripravaka te su pokazali otpornost na tri ili više antimikrobnih pripravaka, dok su izolati S. sciuri bili otporni na dva testirana antimikrobna pripravka.
Ključne riječi: vrste Staphylococcus, tovni pilići, otpornost na antimikrobne pripravke, S. aureus, S. simiae, S. sciuri,

