Bacterial composition in relation to organic and inorganic elements and protein content in dog and cat urine
D.Lukačević*, Z. Vidić, S. Katić, T. Maleš, I. Škoko, D. Balić and M. Benić
Damir Lukačević1* (corresponding author), d.lukacevic.vzs@veinst.hr, orcid.org/0000-0002-6923-0205; Zdravka Vidić1, z.romac.vzs@veinst.hr, orcid.org/0009-0001-7988-2303; Sanda Katić1, s.katic.vzs@veinst.hr, orcid.org/0009-0001-4095-2984; Toni Maleš1, tmales.vzs@veinst.hr, orcid.org/0000-0001-8638-5551; Ines Škoko2, i.skoko.vzs@veinst.hr, orcid.org/0009-0004-0166-9803; Davor Balić3, balic@veinst.hr, orcid.org/0000-0002-7515-0092; Miroslav Benić4, benic@veinst.hr; orcid.org/0000-0001-7594-520X.
1 Laboratory for Diagnostics, Veterinary Department Split, Croatian Veterinary Institute, 21000 Split, Croatia
2 Laboratory for Microbiology and Analytical Chemistry, Veterinary Department Split, Croatian Veterinary Institute, 21000 Split, Croatia
3 Laboratory for Diagnostics, Veterinary Department Vinkovci, Croatian Veterinary Institute, 32100 Vinkovci, Croatia
4 Laboratory for Mastitis and Row Milk Quality, Department of Bacteriology and Parasitology, Croatian Veterinary Institute, 10000 Zagreb, Croatia
https://doi.org/10.46419/cvj.57.4.2
Abstract
Clinical signs of lower urinary tract inflammation are common in domestic carnivores. In dogs, they are usually the result of a bacterial infection, while in cats, they are more often caused by sterile inflammation or behavioural disorders. The prevalence of individual bacterial pathogens isolated from urine samples of dogs and cats was determined, and the relationship between a positive urine culture and the presence of organic and inorganic elements, and proteins was investigated. From January 2022 to June 2024, a total of 563 dog urine samples and 86 cat urine samples were analysed in the Split branch of the Laboratory for Diagnostics, Croatian Veterinary Institute. A clinically significant number of bacteria were isolated from 196 of 563 (34.81%) dog urine samples and 21 of 86 (24.41%) cat urine samples. Of the total number of bacterial isolates, 141 (64.4%) were gram-negative and 78 (35.6%) were gram-positive. Escherichia coli was the most common isolate (45.9% in dogs, 33.3% in cats). In dogs, the second most common isolates were Staphylococcus spp. (20.9%), Streptococcus spp. (9.2%), Proteus spp. (7.7%), Enterococcus spp. (4.6%), Klebsiella spp. (4.08%), and Pseudomonas spp. (3.6%), while other bacterial isolates were <2%. In cats, the most frequently isolated bacteria were Proteus spp. (23.8%), Pseudomonas spp. (14.3%), Streptococcus and Enterococcus spp. (9.5% each), and Staphylococcus spp. and Pasteurella spp. (4.8% each). In dogs, a significant and positive correlation was found between a positive bacteriological result and pyuria, haematuria, proteinuria, and age. In cats, no significant correlations were found between a positive bacteriological culture and variables such as pyuria, haematuria, proteinuria, crystals, or animal age or sex.
Keywords: urine, dog, cat, uropathogen, isolate, bacteria
Introduction
Clinical signs of lower urinary tract inflammation (pollakiuria, periuria, dysuria, stranguria, and haematuria) are common in domestic carnivores. In dogs, these signs are usually the result of bacterial infections, while in cats, they more often stem from sterile inflammatory conditions, such as feline idiopathic cystitis (FIC), urolithiasis, neoplasms, or behavioural disorders. Approximately 14% of dogs develop a bacterial urinary tract infection (UTI) during their lifetime (Ling, 1984), with incidence increasing with age (Passmore, 2007). In cats under 10 years of age, bacterial UTIs are uncommon and affect only 1–8% of the population (Kruger, 1991; Buffington, 1997; Gerber, 2005). They are more common in older cats, in those with chronic kidney disease, and in animals that have undergone invasive urological procedures (Lulich, 1992; Osborne, 1996; Bartges, 2003; Bailiff, 2007). Upper urinary tract infection, or pyelonephritis, is relatively rare in domestic carnivores. UTIs are characterised by colonisation, multiplication and persistence of bacteria within the urinary system, leading to an inflammatory response and the development of clinical signs (Smee & Lloyd, 2013; Weese et al., 2019).
In contrast, subclinical bacteriuria is defined as the presence of bacteria in the urine, confirmed by urine culture, in an animal that shows no clinical signs of urinary tract disease (Weese et al., 2019). The prevalence of subclinical bacteriuria in healthy dogs ranges between 2 and 9% (McGhie, 2014; Wan, 2014; Garcia et al., 2020). A significantly higher prevalence (15–74%) has been found in dogs with diabetes mellitus, obese dogs, puppies with parvoviral enteritis, dogs with acute intervertebral disc extrusion, chronically paralysed dogs, and dogs treated with cyclosporine or glucocorticoids (Koutinas et al., 1998; McGuire et al., 2002; Torres et al., 2005; Lusby et al., 2011; Peterson et al., 2012; Baigi et al., 2017; Olby et al., 2017; Weese et al., 2019). Prevalence rates of 1–13% have been reported in healthy cats (Eggertsdóttir et al., 2011; White et al., 2016; Puchot et al., 2017; Weese et al., 2019). Subclinical bacteriuria differs from bacterial cystitis by the absence of clinical signs, rather than by the number of bacteria. Therefore, a significant bacterial count in the urine (e.g., >100,000 CFU/mL) may be present in clinically healthy animals (Forrester et al., 1999). Subclinical bacteriuria is also not defined by the presence or absence of pyuria. Most UTIs typically result from the ascending entry of bacteria from the external environment or the distal urethra. In the majority of UTIs, a single uropathogenic species is typically isolated. Among gram-negative bacteria, Escherichia coli is the most common causative agent. Among gram-positive bacteria, the most frequently isolated genera are Staphylococcus, Streptococcus, and Enterococcus. Anaerobic bacteria, such as members of the genus Clostridium, are rarely identified as UTI pathogens (Barsanti, 2012). Mixed bacterial infections are also uncommon (Wooley, 1976; Ling, 2001).
Predisposing factors for bacterial UTIs in dogs include: sex (spayed females), anatomical or functional abnormalities affecting continence or complete bladder emptying such as ectopic ureters (Holt, 1995) and hind limb paresis (Olby, 2017), systemic diseases including diabetes mellitus, hyperadrenocorticism (Forrester, 1999) and hyperparathyroidism (Feldman, 2005), and use of glucocorticoids (Ihrke, 1985). Cats with chronic kidney disease, diabetes mellitus, and hyperthyroidism are also predisposed to UTIs (Bailiff, 2006; Mayer-Roenne, 2007). The most reliable diagnostic method for urinary tract infections is bacteriological examination of urine (quantitative urine culture), as it allows for the identification of the bacteria present and the estimation of their approximate number per millilitre. Growth in pure culture is of particular diagnostic importance, as approximately 80% of urinary tract infections are caused by a single bacterial species, 17% by two species in combination, and only 3% by three species (Allen et al., 1987). Growth of several bacterial species usually indicates that one or more species are contaminants.
Microscopic examination of native urine sediment enables qualitative and quantitative assessment of the presence of erythrocytes, leukocytes, epithelial cells, crystals, casts, microorganisms, and artefacts (Barsanti et al., 2004). It provides additional information that supports the interpretation of urine culture results, facilitates the diagnosis of urinary tract infections, assists in sample quality assessment, and guides further diagnostic workup. Occasionally, no leukocytes are detected in the sediment despite the presence of inflammation. This typically occurs in immunocompromised patients, particularly those with diabetes mellitus or undergoing immunosuppressive therapy such as long-term glucocorticoid use, where an inadequate inflammatory response leads to reduced production of inflammatory cells.
The presence of proteins in urine (proteinuria) is determined as part of the overall examination. Depending on the origin, proteinuria can be classified as prerenal, renal, or postrenal, indicating that proteins in the urine can originate from the blood plasma, urinary tract, or reproductive tract. Under normal conditions, urine contains only very small amounts of protein. The presence of significant amounts or abnormal types of proteins is an indicator of renal or systemic disease. The clinician must assess the persistence, origin, and severity of proteinuria, and interpret the findings in the context of other clinical and laboratory results. Physical and chemical urinalysis is usually performed by clinicians during clinical examination of the animal. Physical examination of urine includes the assessment of colour, turbidity, and odour, as well as the measurement of specific gravity using a refractometer. Chemical analysis is carried out using urine test strips, which semi-quantitatively determine the presence of glucose, ketones, nitrites, pH, erythrocytes (haemoglobin), leukocytes (leukocyte esterase), bilirubin, urobilinogen, creatinine, and total protein. The aim of laboratory testing is to provide clinicians with additional relevant information to aid in diagnosis and to guide treatment decisions or further diagnostic procedures.
In addition to bacterial infections, as the most common cause of urinary tract disease, there are also non-infectious pathological processes such as neoplasia or urolithiasis that can trigger an inflammatory response accompanied by the exudation of erythrocytes, leukocytes, and proteins into the urine. The presence of blood cells and proteins in urine is indicative of an inflammatory process within the urinary tract, but does not provide information about its aetiology or anatomical location. Although the microscopic detection of bacteria in fresh urine sediment may raise suspicion of a UTI, the diagnosis should always be confirmed by urine culture.
Cystocentesis is the recommended method for collecting samples for urine culture, as it bypasses the distal urethra, which is colonised with commensal bacteria even in healthy dogs (Ling & Ruby, 1978; Weese et al., 2019). This sampling method eliminates the issue of distinguishing contaminants from true uropathogens. If dysuria and/or pollakiuria make cystocentesis difficult or impossible, there are alternative collection methods, such as free-catch urine, manual bladder expression, or catheterisation. In such cases, the external genitalia must be cleaned with a suitable solution before sampling. In long-haired breeds, the hair around the vulva should be clipped. Since a urethral catheter provides direct access between the external environment and the urinary bladder, catheterisation always carries a risk of bacteriuria or bacterial cystitis. Catheters must be sterile and in impeccable condition to avoid iatrogenic UTIs.
The aim of this study was to present the prevalence of individual uropathogens isolated from the urine samples of dogs and cats at the Laboratory for Diagnostics, Croatian Veterinary Institute – Split Branch, during the period from January 2022 to June 2024, and to investigate the relationship between positive urine culture results and the presence of organic and inorganic elements, and proteins in urine.
Materials and methods
A retrospective analysis was performed on the results of laboratory examinations of urine samples from dogs and cats collected between January 2022 and June 2024. During this period, 563 dog urine samples and 86 cat urine samples were examined. The samples were submitted by small animal veterinary clinics, primarily from the Split area, and were obtained by free-catch, catheterisation, or cystocentesis. Most samples were submitted due to clinical suspicion of a UTI, while a smaller number were provided as control samples. For each sample, information regarding the species, breed, sex, and age of the animal, and method of collection (when specified) were recorded. Routine urine analysis in our laboratory includes quantitative bacteriological culture and protein detection. Microscopic examination of native urine sediment was performed upon request, which applied to the majority of samples. Accordingly, we microscopically examined 408 of 563 (72.5%) dog urine samples and 62 of 86 (72.1%) cat urine samples.
Urine was cultured quantitatively, as estimating the number of bacteria per millilitre helps distinguish significant bacteriuria from contamination with periurethral bacterial flora. Samples were collected prior to the initiation of antimicrobial therapy.
Urine inoculation is performed using a calibrated sterile, disposable 10 μL loops. A 10 μL loop delivers 0.01 mL of urine, providing a detection threshold of 10² CFU/mL, while the countable range of colonies (100–999 colonies per plate) corresponds to 10⁴ CFU/mL. (Aspevall et al., 2001). Above this concentration, confluent growth prevents accurate quantification.
There is no single culture medium that allows both the identification and quantification of all bacteria potentially present in urine. Therefore, each sample is routinely inoculated onto blood agar, MacConkey agar, Pseudomonas agar, and UriSelect™4 chromogenic agar. When selecting culture media, it is important to inhibit the swarming of Proteus spp. and to enable the detection of E. coli, which is facilitated by the chromogenic medium UriSelect™4. UriSelect™4 is a non-selective chromogenic medium for the isolation and enumeration of bacteria. It enables the direct identification of the most common pathogens of urinary tract infections (Escherichia coli, Proteus mirabilis, and Enterococcus spp.) through the demonstration of their enzymatic activity. In addition, it allows for presumptive identification of several other uropathogens, particularly from the KESC group of Enterobacteriaceae (Klebsiella, Enterobacter, Serratia, and Citrobacter) and the PMP group (Proteus-Morganella-Providencia).
After incubation under aerobic conditions at 37°C for 18–24 hours, plates were visually examined to assess bacterial growth, colony morphology, and the approximate number of colonies (CFU; colony-forming units) per millilitre of urine (CFU/mL). The number of colonies for each morphotype observed in the culture was determined separately. Samples were considered positive if they contained ≥10⁴ colony-forming units (CFU)/mL for catheterised or voided urine, and/or ≥10³ CFU/mL for cystocentesis samples. It was also defined as the growth of no more than two potential uropathogens (Sørensen et al., 2016). The growth of more than two bacterial morphotypes was considered indicative of contamination. In addition to the colony count (CFU/mL), the isolation of common uropathogens such as Enterobacterales or coagulase-positive Staphylococcus species was used as a basis for evaluating bacteriological culture results. Bacterial identification was performed using the KOH test, catalase production and oxidase activity, following the guidelines of Markey et al. (2013).
Most bacteria were identified to the genus level. In the case of Enterobacteria and certain other gram-negative bacteria, isolates were identified to the species level using a commercial biochemical identification system (API 20E, BioMérieux, France).
Microscopic examination of native urine sediment
Sample collection and handling
Standardised sample preparation for microscopic examination of urine sediment is essential to obtain objective and consistent results, as variations in centrifugation speed or duration can influence results. Other technical variables, such as sample storage time and temperature, may also affect the results. At room temperature, cell and cast degradation begins within 2 hours of urine collection, while refrigeration can lead to precipitation of amorphous crystals. In the standardised procedure, a defined urine volume (approximately 5 mL) is centrifuged for 5 minutes at 400 × g (relative centrifugal force) or at 1500–2000 rpm (rotations per minute). A drop of the resulting sediment (about 10 μL) is then examined microscopically under 400× magnification (high-power field). At least 10 fields of view are assessed, and the average number of leukocytes, erythrocytes, and other elements per field is recorded. Haematuria is defined as the presence of more than five erythrocytes, and significant pyuria as more than five leukocytes per high-power field (400×) of urine sediment (Osborne & Stevens, 1999). An active urinary sediment is characterised by the presence of pyuria and/or haematuria.
Sulfosalicylic acid test for protein detection
The test is performed exclusively on samples collected prior to the administration of diagnostic or therapeutic agents. The supernatant of centrifuged urine samples is used to avoid false-positive results caused by proteinaceous material typically present in the sediment (e.g., erythrocytes, leukocytes, epithelial cells, renal casts; Osborne & Stevens, 1999). A 20% solution of sulfosalicylic acid (SSA) is used to precipitate all dissolved proteins, including albumins, glycoproteins, globulins, Bence-Jones proteins, and haemoglobin. The protein concentration is then estimated based on the degree of sample turbidity. The test is performed by adding 5–10 drops of 20% SSA to approximately 5 mL urine. Depending on the amount of protein present, a corresponding level of turbidity will develop. A control test tube containing only the untreated urine sample is observed alongside the test tube with SSA to allow for visual comparison.
SSA test results are interpreted as follows:
- a) No turbidity – Negative reaction (equivalent to <0.1 g/L)
- b) Trace protein – Slight turbidity visible against a dark background (corresponds to approximately 0.1 g/L)
- c) Positive reactions:
1+ = Slight turbidity (0.1–0.5 g/L)
2+ = Moderate turbidity (0.5–2 g/L)
3+ = High turbidity (2–5 g/L)
4+ = Flocculent precipitation (>5 g/L)
The SSA test results are interpreted by experienced laboratory personnel. Since the assessment of turbidity levels (i.e., 1+ to 4+) is not standardised, variability in interpretation may occur between individual analysts and laboratories (Osborne & Stevens, 1999).
Results
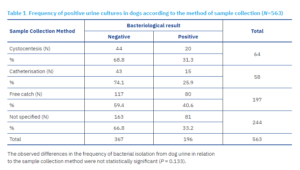
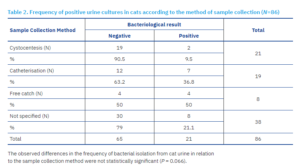
Mid-stream urine collection accounted for 205 of 649 samples (31.6%), including 197 (35%) from dogs and 8 (9.3%) from cats. Cystocentesis was performed in 85 cases (13.1%), comprising 64 (11.4%) dog samples and 21 (24.4%) cat samples. A total of 77 samples (11.9%) were obtained by catheterisation, including 58 (10.3%) from dogs and 19 (22%) from cats. In 282 cases (43.5%), the method of collection was not specified; of these, 244 (43.3%) were dog samples and 38 (44.2%) were cat samples.
A clinically significant number of bacteria was isolated from 196 of 563 (34.81%) dog urine samples and from 21 of 86 (24.41%) cat urine samples. Of the total 217 positive samples, 22 (10.1%) were collected via cystocentesis: 20 (9.2%) from dogs and 2 (0.9%) from cats. A further 22 out of 217 (10.1%) samples were obtained by catheterisation: 15 (6.9%) from dogs and 7 (3.2%) from cats. A total of 84 of 217 (38.7%) samples were collected using the free-catch method: 80 (40.8%) from dogs and 4 (19%) from cats. For a considerable proportion of samples, 89 of 217 (41%), the collection method was not specified by the submitting veterinarians.
Dogs
Cats
Bacterial isolates
Significant bacteriuria was found in 34.8% of dog samples and 24.4% of cat samples. In the vast majority of cases (>99%), a single bacterial culture was isolated, and significant growth of two bacterial morphotypes was detected in only two canine urine samples. Of the total number of bacterial isolates, 141 (64.4%) were gram-negative bacteria and 78 (35.6%) were gram-positive. Escherichia coli was the most frequent isolate (45.9% in dogs and 33.3% in cats). In dogs, this was followed by Staphylococcus spp. (20.9%), Streptococcus spp. (9.2%), Proteus spp. (7.7%), Enterococcus spp. (4.6%), Klebsiella spp. (4.08%), and Pseudomonas spp. (3.6%), while the remaining bacterial isolates were present in significantly lower numbers (<2%). In cats, the next most frequent were Proteus spp. (23.8%), Pseudomonas spp. (14.3%), Streptococcus and Enterococcus spp. (9.5% each), and Staphylococcus spp. and Pasteurella spp. (4.8% each).
Microscopic examination of sediment and protein testing
Only urine samples that underwent microscopic examination of sediment and protein testing were considered for further analysis.
Dogs
The frequency of bacteriologically positive urine culture results in dogs did not differ significantly over the observed years (2022–2024, p = 0.669). Microscopic examination of native urine sediment was performed on 408 of 563 (72.5%) submitted dog urine samples. Of the 196 bacteriologically positive urine samples, 140 also underwent microscopic sediment examination. Of the 367 bacteriologically negative urine samples, 268 were subjected to microscopic sediment examination.
Significant pyuria was present in 94 of 140 (67.1%) dogs with a positive bacteriological finding, and in 44 of 268 (16.4%) of negative cases. Microscopic haematuria was observed in 43 of 140 (30.7%) dogs with a positive bacteriological finding and 55 of 268 (20.5%) negative cases. Crystals were present in 21 of 140 (15%) bacteriologically positive urine samples and in 47 of 268 (17.5%) negative cases.
All submitted samples were routinely tested for the presence of proteins. In total, proteinuria of any intensity was found in 53% of bacteriologically positive samples, where the proteinuria was moderate to pronounced in 35.7% and mild in 17.3% of samples. In bacteriologically negative samples, proteinuria was detected in 27.2% of samples, where it was moderate to marked in 72 of 367 (19.6%), and mild in 28 of 367 (7.6%) samples.
In the subsequent analysis, all samples included were subjected to microscopic sediment examination and protein testing, with the exception of samples lacking sex or age data, resulting in a sample size of 129 bacteriologically positive and 244 bacteriologically negative dogs. Pearson’s correlation coefficient was then calculated to assess the relationship between a positive bacteriological finding, significant pyuria, haematuria, crystalluria, proteinuria, sex, and age in dogs (Table 3).
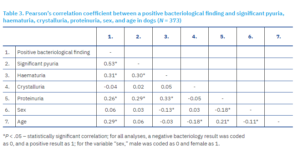
A statistically significant, moderate, positive correlation was found between a positive bacteriological finding and significant pyuria, and significant pyuria was more common in dogs with a positive bacteriological finding. A statistically significant, weak, positive correlation was observed between positive bacteriological findings and both haematuria and proteinuria, which were both more frequent in dogs with a positive bacteriological finding. A statistically significant, weak, positive correlation was also observed between a positive bacteriological finding and age, as older dogs were more likely to have a positive bacteriological finding. No statistically significant correlation was found between positive bacteriological findings and the presence of urinary crystals or animal sex. A statistically significant, weak, positive correlation was observed between the presence of proteinuria and significant pyuria, haematuria, and age, while there was a significant, weak and negative correlation with sex, with proteinuria occurring more frequently in male dogs than in female dogs. No statistically significant correlation was found between proteinuria and the presence of urinary crystals. A statistically significant weak positive correlation was observed between significant pyuria and haematuria. A statistically significant weak negative correlation was found between haematuria and sex, with haematuria being more common in males. No statistically significant correlation was found between the presence of urinary crystals and any of the variables examined, except for age. A statistically significant weak negative correlation was observed, indicating that urinary crystals were more common in younger individuals. Finally, a statistically significant, weak, negative correlation was found between sex and age, as in this sample, younger individuals were more likely to be female.
Independent sample t-tests were calculated to determine differences in the examined variables between bacteriologically negative and positive dogs (Table 4).
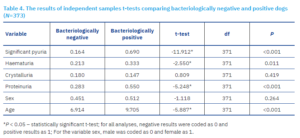
The occurrence of significant pyuria significantly differed between bacteriologically positive and negative dogs, and was more frequently observed in positive dogs. The frequency of haematuria differed between bacteriologically positive and negative dogs, and was more frequent in positive dogs. Additionally, the frequency of proteinuria differed significantly between bacteriologically positive and negative dogs, and was more common in dogs with a positive urine culture. Age also significantly differed between positive and negative urine cultures, as positive dogs were significantly older. No statistically significant differences were found for sex or the presence of urinary crystals between bacteriologically positive and negative dogs.
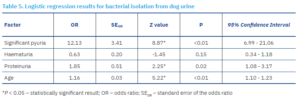
Dogs with pyuria had 12.13 times higher odds of having a positive urine culture (p < 0.01), while dogs with proteinuria had 1.85 times higher odds of a positive urine culture (p = 0.02). With each additional year of age, the odds of a positive urine culture increased by a factor of 1.16 (p < 0.01).
Cats
There were no significant differences (p = 0.892) in the frequency of bacteriologically positive urine cultures during 2022–2024 . Microscopic examination of native urine sediment was performed on 62 of 86 (72.1%) cat urine samples. In total, 21 were bacteriologically and of these 10 (47.6%) underwent microscopic sediment examination, while 65 were bacteriologically negative samples and of these 52 (80%) were examined microscopically.
Significant pyuria was observed in 4 of 10 (40%) microscopically examined positive urine samples, and in 9 of 52 (17.3%) bacteriologically negative samples. Microscopic haematuria was present in 4 of 10 (40%) cats with a positive bacteriological finding, and 20 of 52 (38.5%) bacteriologically negative samples. The presence of urinary crystals was confirmed in 2 of 10 (20%) bacteriologically positive samples and in 9 of 52 (17.3%) negative samples.
All submitted samples were routinely tested for the presence of proteins, and 66.7% of all bacteriologically positive samples. Proteinuria was moderate to marked in 10 of 21 (47.6%) bacteriologically positive samples, and mild in 4 out of 21 (19%) samples. Among bacteriologically negative samples, proteinuria was present in 46.2% of samples, and was moderate to marked in 20 of 65 (30.8%), and mild in 10 of 65 (15.4%) samples.
In the subsequent analysis, microscopic sediment examination and protein testing were performed on samples regardless of bacteriological result, with the exception of samples lacking sex or age data, resulting in a sample size of 9 bacteriologically positive and 44 negative cats. Pearson’s correlation coefficient was calculated to assess the relationship between a positive bacteriological finding, significant pyuria, haematuria, crystalluria, proteinuria, sex, and age in cats (Table 6).
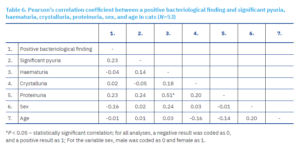
No statistically significant correlations were found among the examined variables, with the exception of the relationship between haematuria and proteinuria, where the correlation was moderate and positive, as a higher protein presence in urine was associated with a higher frequency of haematuria.
Independent samples t-tests were conducted to examine differences in the studied variables between bacteriologically positive and negative cats (Table 7).
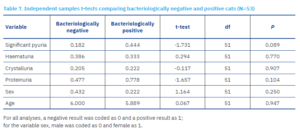
No statistically significant differences were found in the frequency of significant pyuria, haematuria, presence of urinary crystals or proteins, sex, or age between bacteriologically positive and negative cats. This could be explained by the disproportionate ratio of bacteriologically positive to negative cat samples, since only nine cats tested bacteriologically positive, limiting the reliability of the results and reducing the statistical power of the analysis.
Discussion
The quality of the submitted sample is critical for laboratory urine test results to have true clinical relevance in establishing an etiological diagnosis. Improper sampling, storage, or transport of urine can affect the degree of bacteriuria (1–3). Contamination of the sample with periurethral microflora may lead to false-positive findings, potentially resulting in inappropriate treatment and contributing to antibiotic overuse. The term significant bacteriuria was introduced in human medicine in the 1950s with the aim of making urine obtained from midstream specimens comparable to urine collected via suprapubic aspiration (cystocentesis), a method that avoids contamination from periurethral microflora. The original threshold value of ≥10⁵ CFU/mL—also adopted in veterinary medicine—has since been lowered and adjusted to different clinical presentations of UTIs. However, lowering the threshold increases the sensitivity of bacteriological urine testing while reducing specificity, meaning there is a higher probability that the isolated microorganisms represent contamination rather than infection (Tonkić et al., 2017).
In this study, a significantly higher number of dog urine samples (86.7%) were analysed compared to cat samples (13.3%). A similar trend was observed in a study by Fonseca et al. (2021), which also reported a higher number of samples originating from dogs (66.9%) than cats (33.1%). This difference may be explained by a higher prevalence of urinary tract infections in dogs or by the relative ease of obtaining urine samples from dogs (Buffington et al., 1997; Rampacci et al., 2018; Fonseca et al., 2021; Garcês et al., 2022). It may also be associated with the generally higher number of dogs kept as pets compared to cats (Murray et al., 2010).
The results of microscopic urine sediment examination should be interpreted in the context of other clinical data. While microscopic findings are useful in evaluating potential urinary tract diseases, they are not highly predictive of bacteriological culture results and do not correlate with clinical signs of upper or lower urinary tract disease. Pyuria, haematuria, and proteinuria may accompany various inflammatory conditions of the urinary tract not caused by bacterial infection (Bartges, 2004). Accordingly, inflammation of the urinary tract is by no means synonymous with urinary tract infection. Some studies have confirmed a correlation between an increased leukocyte count in the sediment and a higher likelihood of a positive bacteriological culture (Forrester et al., 1999; O’Neil et al., 2013; Lippi et al., 2022), though this correlation has not been consistently confirmed across all studies (McGuire et al., 2002; Swenson et al., 2004). An increased number of erythrocytes in the urine sediment is also not a predictor of a positive bacteriological result (Forrester et al., 1999; O’Neil et al., 2013; Weese et al., 2019).
Postrenal proteinuria is typically considered an accompanying feature of bacterial cystitis and other lower urinary tract inflammations (Bagley et al., 1991). However, this theory has not been supported by other studies. The lack of association between a positive urine culture and proteinuria has been reported by Meindl et al. (2019) and Strachan et al. (2022), who argued that proteinuria is not a consequence of a positive bacteriological finding but rather the result of other, unidentified causes. In the absence of signs of inflammation or bleeding, proteinuria is generally considered to be of renal origin (Lees et al., 2005). However, in cases with clear signs of inflammation, bleeding, and a positive urine culture, determining the origin of proteinuria is more complex, as it may be renal, postrenal, or both.
Dogs
Escherichia coli was the most frequent isolate, followed by Staphylococcus spp. (20.9%), Streptococcus spp. (9.2%), Proteus spp. (7.7%), Enterococcus spp. (4.6%), Klebsiella spp. (4.08%), and Pseudomonas spp. (3.6%), while other isolates were detected in considerably lower numbers (<2%). A similar distribution of bacterial pathogens has been reported elsewhere (Wong et al., 2015; Fonseca et al., 2021; Garcês et al., 2022; Ataya et al., 2023). Other bacterial species isolated sporadically have also been reported by other authors (Budreckis et al., 2015; Wong et al., 2015; Dupont et al., 2020; Gómez-Beltrán et al., 2020; Fonseca et al., 2021; Garcês et al., 2022; Smoglica et al., 2022).
In the present study, a significant and moderate positive correlation was found between a positive bacteriological result and significant pyuria, which is consistent with findings from other studies (Forrester et al., 1999; O’Neil et al., 2013; Lippi et al., 2022). A higher frequency of haematuria was also observed in bacteriologically positive samples, although haematuria can also occur iatrogenically following cystocentesis or catheterisation. Pathological causes include bleeding in the urinary tract due to trauma, coagulopathy, inflammation, infection, necrosis, or neoplasia (Callens and Bartges, 2015; Vap and Shropshire, 2017).
Proteinuria was confirmed in 53% of bacteriologically positive dog urine samples, which is somewhat higher than the report of proteinuria in 42% of positive samples by Fulton et al. (2023). A significant though weak positive correlation was found between positive bacteriological results and proteinuria and between proteinuria and significant pyuria, haematuria, and age. These findings counter Vaden et al. (2004), who found that pyuria is rarely associated with proteinuria. Bagley et al. (1991) and Vaden et al. (2004) suggested that haematuria is unlikely to cause proteinuria, particularly when haematuria is not marked, though they acknowledged that more pronounced haematuria may contribute to the presence of proteinuria. The higher frequency of proteinuria in older dogs may be explained by more frequent glomerular damage leading to impaired filtration and protein balance. Chronic diseases such as diabetes, hypertension, or heart disease can compromise renal function and contribute to the development of proteinuria.
No statistically significant correlation was found between positive bacteriological results and the presence of urinary crystals or sex. Crystals were present in only 15% of bacteriologically positive samples, and the percentage was slightly higher only in samples containing Staphylococcus and Proteus species, both known to produce the enzyme urease. These bacteria create favourable conditions for the formation of struvite crystals (Biegen et al., 2013; Raab et al., 2015; Weese et al., 2019).
Cats
The prevalence of urinary tract infections in cats is generally low. According to studies (Lekcharoensuk et al., 2001; Kruger et al., 2009), it ranges from 1 to 3% in young and middle-aged cats. The feline urinary tract is typically considered an unfavourable environment for microbial colonisation. Among cats diagnosed with lower urinary tract disease, the prevalence of UTIs has been reported to range from 4.9% to 22% (Kraijer et al., 2003; Gerber et al., 2005; Eggertsdottir et al., 2007; Bailiff et al., 2008; Dorsch et al., 2014; Lew-Kojrys et al., 2017). A substantially higher proportion of positive urine cultures (38.5%) was reported by Habrun et al. (2011). In the present study, approximately one-quarter of the analysed feline urine samples were bacteriologically positive. The considerable variation in reported UTI prevalence across studies may be attributed to differences in sampling methods and inconsistent diagnostic criteria, such as the inclusion of free-catch samples and variation in the definition of clinically significant bacteriuria or CFU/mL thresholds.
In the present study, the most frequently isolated bacterial species was Escherichia coli (33.3%), which is consistent with findings reported by numerous other authors (Norris et al., 2000; Ling et al., 2001; Prescott et al., 2002; Ball et al., 2008; Hall et al., 2013; Windahl et al., 2014; Wong et al., 2015; Marques et al., 2016; McMeekin et al., 2017; Moyaert et al., 2017; Fonseca et al., 2021; Garcês et al., 2022). This was followed in frequency by Proteus spp. (23.8%), Pseudomonas spp. (14.3%), Streptococcus spp. and Enterococcus spp. (each 9.5%), and Staphylococcus spp. and Pasteurella spp. (each 4.8%). A similar distribution of bacterial pathogens has been documented elsewhere (Wong et al., 2015; Fonseca et al., 2021; Garcês et al., 2022; Ataya et al., 2023).
In this study, no significant correlation was found between a positive bacteriological culture result and the presence of significant pyuria. Pyuria is commonly observed in patients with subclinical bacteriuria (White et al., 2016; Puchot et al., 2017; Weese et al., 2019) and is frequently detected in cats with idiopathic cystitis (Lund et al., 2013). Similarly, an increased number of erythrocytes in the urine sediment is not considered a reliable predictor of a positive bacteriological culture (Forrester et al., 1999; O’Neil et al., 2013; Weese et al., 2019). This study confirmed such findings, as no significant correlations were observed between haematuria and a positive bacteriological result. Kovarikova et al. (2020) also reported no significant correlation between haematuria and specific lower urinary tract conditions in cats, including feline idiopathic cystitis, urolithiasis, UTIs, or urethral plug obstruction. Furthermore, no significant correlations were found between positive bacteriological results and proteinuria. This aligns with findings by Meindl et al. (2019) and Strachan et al. (2022), who proposed that proteinuria is more likely attributable to other, as yet unidentified, causes rather than to bacteriuria. In this study, proteinuria was observed in 66.7% of bacteriologically positive samples and in 46.2% negative samples. Urinary crystals were detected in 20% of bacteriologically positive samples, though the correlation was not significant. Additionally, no significant correlations were found between bacteriological results and sex.
References [… show]
Prevalencija pojedinih bakterijskih uropatogena izdvojenih iz uzoraka urina pasa i mačaka te povezanost pozitivnih bakterioloških nalaza sa prisutnosti organskih i anorganskih elemenata kao i bjelančevina u urinu.
Damir Lukačević1* (dopisni autor), d.lukacevic.vzs@veinst.hr, orcid.org/0000-0002-6923-0205; Zdravka Vidić1, z.romac.vzs@veinst.hr, orcid.org/0009-0001-7988-2303; Sanda Katić1, s.katic.vzs@veinst.hr, orcid.org/0009-0001-4095-2984; Toni Maleš1, tmales.vzs@veinst.hr, orcid.org/0000-0001-8638-5551; Ines Škoko2, i.skoko.vzs@veinst.hr, orcid.org/0009-0004-0166-9803; Davor Balić3, balic@veinst.hr, orcid.org/0000-0002-7515-0092; Miroslav Benić4, benic@veinst.hr; orcid.org/0000-0001-7594-520X.
1 Laboratorij za dijagnostiku, Veterinarski zavod Split, Hrvatski veterinarski institut, 21000 Split, Hrvatska
2 Laboratorij za mikrobiologiju i analitičku kemiju, Veterinarski zavod Split, Hrvatski veterinarski institut, 21000 Split, Hrvatska
3 Laboratorij za dijagnostiku, Veterinarski zavod Vinkovci, Hrvatski veterinarski institut, 32100 Vinkovci, Hrvatska
4 Laboratorij za mastitise i kakvoću sirovog mlijeka, Odjel za bakteriologiju i parazitologiju, Hrvatski veterinarski institut, 10000 Zagreb, Hrvatska
Sažetak
Klinički znakovi upale donjeg mokraćnog sustava česta su pojava kod domaćih mesojeda. Kod pasa se najčešće javljaju kao posljedice bakterijskih infekcija, dok se kod mačaka češće radi o posljedicama sterilnih upalnih stanja ili poremećaja ponašanja. Cilj ovog rada je prikazati prevalenciju pojedinih bakterijskih uzročnika (uropatogena) izdvojenih iz uzoraka urina pasa i mačaka te istražiti povezanost pozitivnih bakterioloških nalaza sa prisutnosti organskih i anorganskih elemenata kao i bjelančevina. U razdoblju od siječnja 2022. do lipnja 2024. godine, u Laboratoriju za dijagnostiku Veterinarskog zavoda Split, pretraženo je 563 uzoraka urina pasa i 86 uzoraka urina mačaka. Iz 196/563 (34,81%) uzoraka urina pasa i 21/86 (24,41%) uzorka urina mačaka izdvojena je klinički značajna količina bakterija. Od ukupnog broja bakterijskih izolata, gram-negativnih bakterija bilo je 141 (64,4 %), a gram-pozitivnih 78 (35,6 %). Escherichia coli bila je najučestaliji izolat (45,9% u pasa i 33,3% u mačaka). Sljedeći po učestalosti u pasa bili su Staphylococcus spp (20,9%), Streptococcus spp (9,2%), Proteus spp (7,7%), Enterococcus spp (4,6%), Klebsiella spp (4,08%) i Pseudomonas spp (3,6%). Preostali bakterijski izolati bili su zastupljeni u znatno manjem broju (<2%). U mačaka su prema učestalosti slijedili Proteus spp (23,8%), Pseudomonas spp (14,3%), Streptococcus i Enterococcus spp (po 9,5%), te Staphylococcus spp i Pasteurella spp (po 4,8%). U uzorcima urina pasa utvrđena je statistički značajna umjerena pozitivna povezanost između pozitivnog bakteriološkog nalaza i značajne piurije. Utvrđena je statistički značajna slaba pozitivna povezanost između pozitivnih bakterioloških nalaza i hematurije, proteinurije te starosti životinje. U mačaka nije utvrđena statistički značajna povezanost između pozitivnog bakteriološkog nalaza i svih ispitanih varijabli.
Ključne riječi: urin, pas, mačka, uropatogen, izolat, bakterije

