First description of antifungal susceptibility of yeast-like fungi in dermal lesions of dogs in Portugal: A 6-year pilot retrospective study (2018-2023)

R. Lopes*, A. Garcês, A. Silva, P. Brilhante-Simões, Â. Martins, L. Cardoso, E. L. Duarte and A. C. Coelho
Abstract
Yeast infections, particularly those caused by species of the genera Malassezia and Candida, are important contributors to otitis and dermatitis in dogs.
This study investigates the prevalence and antifungal resistance of various yeast-like fungi isolated from canine dermal lesions in Portugal. Between 2018 and 2023, samples from dogs with clinically suspected yeast infections were collected and analysed using the Vitek 2 Compact® (bioMérieux, Marcy-l’Étoile, France) and FUNGITEST™ 60780 kit (Bio-Rad, Hercules, USA). A total of 24 fungal isolates were identified, including Malassezia pachydermatis and Candida albicans. Susceptibility testing revealed high sensitivity to antifungal agents such as fluconazole, ketoconazole, itraconazole, and miconazole across most species, with notable resistance observed in Rhodotorula mucilaginosa to multiple antifungals. Intermediate resistance levels were detected in Malassezia spp. to flucytosine and several other antifungals. These findings underscore the need for continuous monitoring and prudent antifungal use to prevent resistance development. The study also highlights the importance of revisiting treatment protocols for Malassezia infections in dogs, given the observed resistance patterns. Further research is recommended to explore the genetic and molecular mechanisms underlying antifungal resistance and develop new therapeutic agents.Key words: antifungal resistance; clinical pathology; dermatology; fungi; infections; mycology; one health; planetary health; yeast-like fungi
Introduction
Yeasts play an important role in veterinary medicine, primarily due to their applications in animal nutrition and health (Patterson et al., 2023; Sampathet et al., 2023). As probiotics, certain yeast strains, like Saccharomyces cerevisiae, enhance the gut flora, improving digestion and nutrient absorption in livestock and pets. They contribute to better growth rates, feed efficiency, and overall health. Additionally, yeast derivatives, such as beta-glucans and mannans, bolster the immune system, providing resilience against infections (Boender et al., 2009; Alugongo et al., 2017; Samidjan et al., 2022).
Not all yeasts are beneficial. Some, particularly species of the genera Malassezia and Candida, are the aetiological agents of otitis and dermatitis, common diseases among dogs and cats. These yeast species are commensal organisms found on the skin and in the ears of dogs and cats. Under normal conditions, they coexist harmlessly with their host, but they can proliferate and cause infection when there is a disruption in the local environment, such as excess moisture, allergies, endocrine disorders, or immunosuppression. Environmental factors, like humid climates and poor ear hygiene, can also promote yeast overgrowth, leading to otitis (Cafarchia et al., 2005; Bajwa, 2019; Guillot and Bond, 2020; Cabañes, 2021).
These infections, while commonly encountered in veterinary practice, present substantial challenges due to their increasing resistance to antifungal treatments. Resistance mechanisms include alterations in drug target sites, efflux pump overexpression, and biofilm formation, which collectively diminish the efficacy of antifungal treatments. Additionally, improper administration of treatment by pet owners can contribute to this issue (Yurayart et al., 2013; Cowen et al., 2015; Robbins et al., 2017; Revie et al., 2018).
Due to the prevalence of these infections, medicated shampoos containing miconazole or ketoconazole, formulated at the appropriate pH, are commonly used to treat yeast and secondary bacterial infections like those involving Staphylococcus spp. For severe yeast infections, itraconazole is the preferred systemic treatment (Korting and Schöllmann, 2009). This lipid-soluble antifungal drug targets skin lipids where yeast thrives, and it is effective at lower doses (3–5 mg/kg once daily with food), if given in concert with medicated shampoos, compared to doses used for invasive fungal infections like cryptococcosis (Rochette et al., 2003; Malik and Krockenberger, 2017). Terbinafine is also sometimes used, though most dermatologists favour itraconazole. For Malassezia otitis externa, topical treatments containing miconazole are typically recommended. Finally, environmental decontamination is crucial, particularly in multi-pet households, to control the spread of fungal infections (Foy and Trepanier, 2010).
The frequent administration of antifungal agents such as fluconazole, ketoconazole, itraconazole, and miconazole has historically managed these infections effectively (Moraru et al., 2019; Guillot and Bond, 2020; Nguyen Trung et al., 2024). However, the rise in resistance, often a consequence of extensive and sometimes inappropriate use of these drugs, has considerably compromised their efficacy. This phenomenon mirrors trends observed in human healthcare, where antifungal resistance has become a critical issue, underscoring the interconnected nature of health challenges across species. This resistance complicates the management of yeast infections in animals, leading to prolonged illness and increased veterinary costs (Fisher et al., 2022; Hui et al., 2024; McCormick and Ghannoum, 2024).
Understanding the prevalence and resistance patterns of fungal species in dermal lesions is crucial for developing effective treatment protocols (Hui et al., 2024). To the authors’ knowledge, no studies in Portugal have thoroughly described the antifungal susceptibility of yeast-like fungi in canine dermal lesions.
In the present study, the pilot analysis of antifungal susceptibility in this context is presented, marking the first work of its kind Portugal.
In this study, we aimed to explore the epidemiology and antifungal susceptibility of fungal species isolated from canine dermal lesions, and to provide a comprehensive overview that can inform both clinical practice and future research.
Materials and methods
Data collection, sampling and diagnostic procedures
Samples from domestic dogs with suspected yeast infections were submitted to INNO Veterinary Laboratories (Braga, Portugal) from 2018 to 2023. Samples (n = 24) were submitted from veterinary practices, including clinics and hospitals, across all geographical regions of mainland Portugal. Each sample included a laboratory requisition form with the relevant signalment and clinical information such as breed, sex, age, vaccination and prophylactic status, clinical signs/clinical suspicion and requested analyses.
Smears from wounds, mucosa (i.e., mouth, anus, vagina), plucked hairs or scraped skin of each animal were collected from the lesions using a sterile lancet by veterinary practitioners and placed in sterile containers. Samples were plated on Sabouraud Gentamicin Chloramphenicol 2 agar (bioMérieux, Marcy-l’Étoile, France) and, incubated at 30°C for 48–72 hours (Figure 1).
Yeast colonies were identified with the yeast identification card (YST ID, REF 21343) on Vitek 2 Compact® (bioMérieux, Marcy-l’Étoile, France).
Following the manufacturer’s recommendations, the antifungigram was performed with the FUNGITEST™ 60780 kit (Bio-Rad, Hercules, USA) (Figure 2).
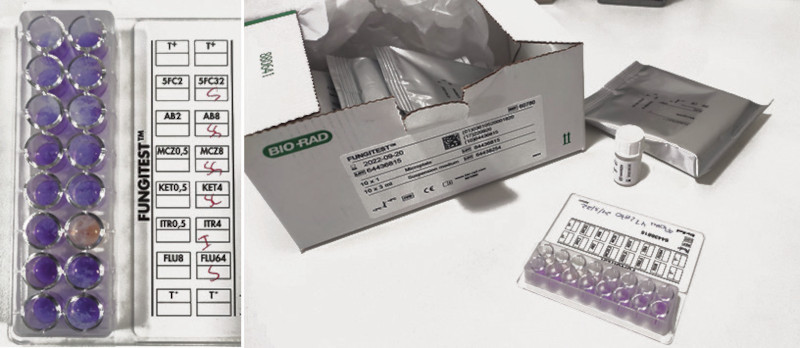
The antifungal agents tested with this kit are 5-fluorocytosine, amphotericin B, miconazole, ketoconazole, itraconazole and fluconazole. The results were interpreted according to the colour of the two wells for each antifungal agent. If both stained blue, it was determined that there was no growth (strain inhibited by the antifungal agent in vitro); dark and blue staining meant reduced growth (strain with intermediate sensitivity); both with positive growth (strain not inhibited by the antifungal agent in vitro).
Statistical analysis
All data were made available in digital format in Clinidata® (Clinidata XXI version 5.3.25, Maxdata Software, S.A., Carregado, Portugal) and transferred to Microsoft Excel® (Microsoft, Redmond, WA, USA) sheets. Statistical analysis was conducted using JMP® (version 14.3 SAS Institute, Cary, NC, 1989-2023 SAS), DATAtab® (DATAtab: Online Statistics Calculator. DATAtab e.U. Graz, Austria, 2024), and MedCalc® Statistical Software (version 20.006, MedCalc Software Ltd, Ostend, Belgium, 2021). For the specific analysis of antifungal susceptibility of fungal species isolated from canine dermal lesions, descriptive statistics were used to calculate the frequencies and percentages of susceptibility, intermediate response, and resistance for each fungal species and antifungal agent. These results were tabulated and compared to identify significant patterns and discrepancies. The percentages of susceptible, intermediate, or resistant samples were calculated to enable a clear visual comparison of the data across species and antifungal agents.
Results
Type of samples and yeast species isolated
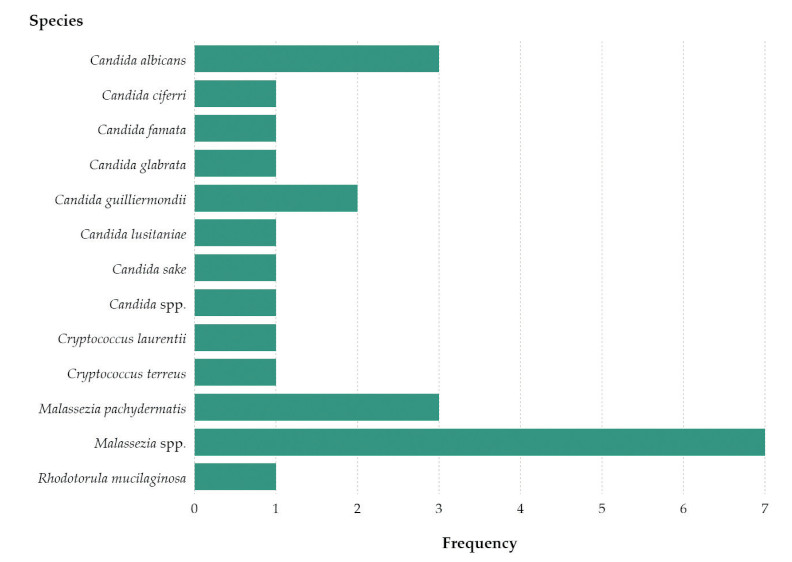
Among the 24 yeast-like fungal isolates tested, the following species were identified: Candida albicans (12.5%; n = 3), Candida ciferri (4.2%; n = 1), Candida famata (4.2%; n = 1), Candida glabrata (4.2%; n = 1), Candida guilliermondii (8.3%; n = 2), Candida lusitaniae (4.2%; n = 1), Candida sake (4.2%; n = 1) and Candida spp. (4.2%; n = 1), Cryptococcus laurentii (4.2%; n = 1), Cryptococcus terreus (4.2%; n = 1), Malassezia pachydermatis (12.5%; n = 3), Malassezia spp. (29.2%; n = 7) and Rhodotorula mucilaginosa (4.2%; n = 1) (Figure 3).
Susceptibility and Resistance Pattern
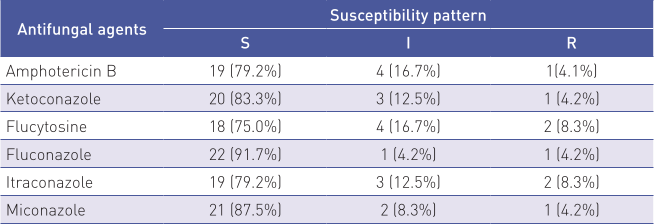
Antifungal susceptibility testing revealed that 19 isolates (79.2%) were susceptible to amphotericin B, 20 (83.3%) to ketoconazole, 18 (75.0%) to flucytosine, 22 (91.7%) to fluconazole, 19 (79.2%) to itraconazole and 21 (87.5%) to miconazole. Higher resistance proportions were noted for flucytosine (8.3%) and itraconazole (8.3%), with lower resistance rates observed for other antifungals. Table 1 presents the susceptibility for each agent.
Analysis of Specific Species
For Candida guilliermondii, both isolates (100%) were susceptible to all antifungal agents tested. Candida albicans showed 100% susceptibility to amphotericin B, ketoconazole, flucytosine, fluconazole and miconazole, while 66.7% were susceptible to itraconazole, with 33.3% showing intermediate susceptibility.
Candida ciferri, Candida famata, Candida glabrata, Candida sake, Cryptococcus laurentii, and Cryptococcus terreus were 100% susceptible to all antifungal agents tested.
For Malassezia pachydermatis, 81.2% were susceptible to amphotericin B and itraconazole, with 18.8% showing intermediate susceptibility. This species was 87.5% susceptible to ketoconazole and miconazole, with 12.5% showing intermediate susceptibility. Additionally, 80.0% were susceptible to flucytosine, with 20.0% resistant. Fluconazole showed 100% susceptibility.
Malassezia spp. demonstrated similar patterns, with 81.2% susceptibility to amphotericin B and itraconazole, 87.5% to ketoconazole and miconazole, 80.0% to flucytosine (with 20.0% resistance), and 100% to fluconazole.
An exception was Rhodotorula mucilaginosa, which exhibited complete resistance to fluconazole and itraconazole, but was 100% susceptible to amphotericin B, ketoconazole, flucytosine, and miconazole.
Discussion
Infections caused by yeasts are very common in veterinary practice, sometimes very difficult to treat, and are recurrent, with high costs for the owner and a high health impact on the animals (Ganguly and Yadav, 2017; Bond et al., 2020; Hui et al., 2024). The present study provides the first insights into the antifungal susceptibility of yeast-like fungi isolated from canine dermal lesions in Portugal.
Only data regarding dogs were included since the data available on cats were almost non-existent. Although cats also can be affected, it seems that dogs are more susceptible to yeast infections, explaining the higher number of these species. Certain dog breeds (i.e., Basset Hounds, Cocker Spaniels, and West Highland White Terriers) are genetically predisposed to skin conditions that promote yeast overgrowth, such as seborrhoea or allergies (Mauldin et al., 1997; Mazrier et al., 2016; Favrot et al., 2020). Dogs have deep ear canals and areas of the skin that can easily trap moisture and create an ideal environment for yeast growth. This is particularly true for breeds with floppy ears (i.e., Cocker Spaniels) or skin folds, which allow Malassezia spp. and other yeast species to thrive (Staroniewicz et al., 2008). Many breeds are often diagnosed with underlying health issues like atopy (allergies) or hormonal disorders (hypothyroidism, Cushing’s disease), which can compromise skin defences and contribute to yeast infections (Alves et al., 2023). Compared to cats, dogs may not groom themselves as thoroughly as cats, leading to the accumulation of debris, moisture, and sebum, which can promote yeast growth. Cats are typically more meticulous groomers, reducing the likelihood of conditions that favour yeast overgrowth. Also, they are often exposed to a wider range of environments (parks, beaches, etc.) and activities (swimming, digging) that can lead to increased moisture and contact with potential sources of infection (Seyedmousavi et al., 2018).
Among the isolated species, the most common were Candida albicans (12.5%; n = 3), Malassezia pachydermatis (12.5%; n = 3) and Malassezia spp. (29.2%; n = 7). This is in correlation with the literature where Malassezia spp. is the most frequently isolated species from dogs (Girão et al., 2006). Species from the genus Candida, particularly C. albicans, are also regarded as significant (Nardoni et al., 2005; Brito et al., 2009).
Antifungal resistance is an emerging concern in veterinary medicine, particularly in the treatment of fungal infections in companion animals like dogs and cats.
While antifungal resistance is less common than bacterial resistance, it presents significant challenges, particularly in recurrent or chronic infections (Arastehfar et al., 2020; Fisher et al., 2022). The primary fungi of concern in companion animals include Malassezia spp., Aspergillus spp., and dermatophytes, with resistance primarily associated with prolonged or inappropriate antifungal use (Brito et al., 2007; Angileri et al., 2019; Gnat et al., 2021; Lopes et al., 2024).
The antifungal agents tested were those available in the commercial kit.
The majority of those agents are used in veterinary medicine (Kaya et al., 2022).
The high susceptibility values observed for most antifungal agents, particularly fluconazole (91.7%), miconazole (87.5%), and ketoconazole (83.3%) suggest that these drugs remain effective treatment options for many yeast infections in dogs.
The intermediate susceptibility observed in several isolates, particularly Malassezia spp. and M. pachydermatis, indicates that while these antifungals are generally effective, there is a subset of infections where treatment might be less predictable. For example, 18.8% of Malassezia pachydermatis isolates showed intermediate susceptibility to amphotericin B and itraconazole, and 12.5% showed intermediate susceptibility to ketoconazole and miconazole. This intermediate resistance suggests that treatment efficacy may vary, and that higher doses or combination therapies might be necessary in some cases.
The presence of antifungal resistance, although relatively low, is still significant and warrants continuous monitoring. The resistance rates observed in this study, such as 8.3% resistance to flucytosine and itraconazole in M. pachydermatis, highlight the potential for resistance development even in veterinary settings.
Azoles (i.e., itraconazole, fluconazole, ketoconazole) are widely used agents for treating fungal infections due to their broad-spectrum activity. However, resistance can develop through: efflux pump overexpression (fungal cells can overproduce efflux pumps, actively removing the drug from the cell), ergosterol pathway mutations (azoles target the ergosterol synthesis pathway in fungi, and mutations in the target enzyme (lanosterol 14α-demethylase) reduce drug efficacy); and cross-resistance (Wang et al., 2021). In the case of polyenes as amphotericin B, resistance mechanisms are less well understood but may involve changes in the fungal cell membrane (Carolus et al., 2020). While resistance to allylamines (i.e., terbinafine) is less common, it has been reported in cases of dermatophyte infections in both animals and humans. Mutations in the squalene epoxidase enzyme, which terbinafine targets, can reduce its efficacy (Kaya et al., 2022; Gupta et al., 2023).
Resistance to azoles has been reported, particularly in animals with chronic otitis externa or dermatitis, since it is one of the most used agents (Kaya et al., 2022). One study (Kano et al., 2019) investigated ravuconazole (an azole antifungal and active metabolite of fosravuconazole) susceptibility and multi-azole resistance in 13 clinical M. pachydermatis strains from canine origin using a modified Clinical and Laboratory Standards Institute M27-A3 test. The BLAST analysis revealed that the ravuconazole-resistant strain had mutations in the ERG11 gene, encoding Gly at codon 461 and Asp in cytochrome P450. These mutations likely alter the affinity of the ERG11-encoded protein for RVZ compared to ITZ, which may explain their resistance.
Weiler et al. (2013) compared the susceptibility of M. pachydermatis isolates from sick and healthy dogs to azole and polyene antifungals: ketoconazole, fluconazole, itraconazole, voriconazole, clotrimazole, miconazole, nystatin, and amphotericin B. The authors stated that M. pachydermatis isolated from animals with otitis are less sensitive to some antifungal agents than yeasts isolated from animals without otitis. Lyskova et al. (2007) tested M. pachydermatis isolates from dogs with and without otitis externa and demonstrated that all of the isolates exhibited a high susceptibility to all tested antifungal agents except fluconazole, to which 4.4% of the isolates were resistant. Eichenberg et al. (2018) tested a group of 82 M. pachydermatis samples from dogs and cats with the following antifungal agents: ketoconazole, fluconazole and itraconazole.
All isolates were susceptible to itraconazole, 2.4% of isolates were resistant to fluconazole, and 3.7% to ketokonazole. Jesus et al. (2011) induced resistance to fluconazole in vitro. M. pachydermatis isolates can become resistant during the course of the treatment with this antifungal. Fluconazole resistant M. pachydermatis isolates exhibited cross-resistance to other azoles.
Mendes et al. (2017) reported significant antifungal resistance among yeast isolates from various environments, including hospital settings, cow’s milk, and wild animal microbiota. Their study found 20.1% resistance to fluconazole and 12.4% to amphotericin B, underscoring the broader issue of antifungal resistance beyond veterinary contexts.
The results presented here are similar to those described by other authors. Is important to note that in this study, Rhodotorula mucilaginosa exhibited resistance to both fluconazole and itraconazole.
This is expected, since this yeast exhibits intrinsic resistance to several commonly used antifungals, such as azoles and echinocandins, due to its natural cell wall and membrane properties likely to contribute to its reduced azole permeability and overall tolerance, even in the absence of acquired resistance mechanisms, like mutations or efflux pump overexpression (Jarros et al., 2020; Gu et al., 2021).
Despite the value of these findings, one of the primary limitations of this study is that antifungal susceptibility testing is regrettably an infrequently requested analysis. This is largely due to a lack of awareness among veterinary clinicians about the utility of such testing and the existence of antifungal resistance.
The absence of routine susceptibility testing means that the emergence of resistant strains might go unnoticed until treatment failures become evident, which can complicate management strategies and outcomes. There is therefore a significant need to raise awareness among veterinarians about the benefits of susceptibility testing. Increased sensitivity to these issues can lead to improved antimicrobial stewardship and patient outcomes.
Given the variability in susceptibility and the presence of intermediate resistance, relying solely on empirical treatment without susceptibility testing could lead to suboptimal outcomes.
Regular testing allows the identification of emerging resistance patterns and helps in tailoring treatment protocols to ensure maximum efficacy. This approach is particularly crucial for managing infections caused by species like Rhodotorula mucilaginosa, which showed resistance to multiple antifungal agents.
Antifungal resistance is a growing concern in veterinary medicine, particularly in companion animals like dogs and cats. Species of the genera Malassezia and Candida are increasingly showing resistance to commonly used antifungal agents like azole drugs (i.e., fluconazole, itraconazole). The mechanisms of resistance, such as efflux pump overexpression and mutations in target enzymes, are similar to those seen in human pathogens, raising concerns about cross-species transmission and treatment failures (Picot et al., 2022; Gupta et al., 2023). Companion animals, particularly those with chronic or recurrent fungal infections, can act as reservoirs for resistant fungi. Zoonotic transmission can lead to the spread of antifungal-resistant infections (Kundu et al., 2024). For example, Malassezia pachydermatis and Candida spp., which can infect both animals and humans, have been found in shared environments, such as homes or veterinary clinics.
Immunocompromised individuals, such as the elderly or those undergoing chemotherapy, are at higher risk of acquiring fungal infections from resistant strains carried by pets. Companion animals often share environments with humans, and environmental contamination by resistant fungi poses a broader risk (Garcia-Bustos et al., 2023).
Overuse of antifungals in pets, such as the frequent application of topical or systemic antifungal treatments, can select resistant strains in the environment.
These resistant pathogens can persist on surfaces, in grooming areas, or in veterinary hospitals, contributing to the spread of antifungal resistance.
Extremely resistant pathogenic yeasts, such as Candida auris, that pose a serious concern in human hospital settings are not common in veterinary practice. Still, an increase in resistant strains suggests that further research into alternative antifungal therapies is warranted also in veterinary medicine. Investigating novel antifungal agents or alternative treatment strategies, such as combination therapy or the use of biocides, could provide additional tools for managing resistant infections (Gupta et al., 2023).
Moreover, the restriction in use of some antifungals in veterinary medicine such as for antibiotics (i.e., critical important antibiotics) is not in place, although more recent antifungal drugs are seldom used in veterinary medicine due to their cost.
This study highlights the relevance of a One Health approach in understanding and managing antifungal resistance.
The interconnectedness of human, animal, and environmental health means that resistance patterns observed in veterinary medicine could have broader implications. Monitoring antifungal resistance in animals can provide early warnings of potential resistance trends that could impact human health.
Recommendations for the future
Veterinary practitioners need to be educated about the importance of antifungal susceptibility testing, as increased awareness can lead to better understanding and management of fungal infections, thereby improving clinical outcomes. Encouraging the responsible use of antifungal agents is crucial; overuse and misuse can lead to the development of resistance, so veterinarians should adhere to guidelines on appropriate dosing and duration of therapy. Adopting a One Health approach is vital, as understanding the interconnectedness of human, animal, and environmental health can help in monitoring and managing antifungal resistance more comprehensively (Woods et al., 2023). Treatment guidelines for veterinary fungal infections are still missing and could promote the judicious use of antifungal drugs to minimise the development of resistance.
Conclusions
The findings presented here provide a comprehensive overview of the antifungal susceptibility of yeast-like fungi isolated from canine dermal lesions in Portugal. The high susceptibility rates to fluconazole, miconazole, and ketoconazole are promising, indicating that these antifungal agents remain effective treatment options for many yeast infections in dogs. However, the significant resistance observed in Rhodotorula mucilaginosa highlights the necessity for alternative treatments and underscores the critical need for ongoing surveillance.
While high susceptibility rates to several antifungal agents do not point towards an alarming setting such as for some bacterial pathogens, the presence of resistance and intermediate susceptibility in some isolates underscores the need for routine susceptibility testing and prudent antifungal use. By addressing the lack of awareness and incorporating regular testing, the veterinary community can better manage fungal infections and mitigate the risk of resistance development.
Antifungal resistance is an emerging threat with significant implications for both animal and human health. Through a One Health approach, efforts to monitor, prevent, and manage resistance must consider the close relationship between humans, animals, and the environment. Coordinated action is essential to curbing the spread of resistant fungal pathogens and ensuring the effectiveness of antifungal treatments across species.
Funding
This study was supported by projects: UIDB/00772/2020 (doi:10.54499/UIDB/00772/2020), UIDP/00681/2020 (Doi:10.54499/UIDP/00681/2020), LA/P/0059/2020, UIDB/05183/2020 (Doi: 10.54499/UIDB/05183/2020), UIDP/05183/2020 (Doi:10.54499/UIDP/05183/2020)and LA/P/0121/2020 (Doi:10.54499/LA/P/0121/2020), funded by the Portuguese Foundation for Science and Technology (FCT).
Institutional Review Board Statement
All procedures complied with the Portuguese legislation for the protection of animals used for scientific purposes (i.e., Decree-Law no. 113/2013, of 7 August 2013), which transposes European legislation (i.e., Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010). The study project was approved by the Institutional Review Board of INNO Veterinary Laboratories (protocol code INNO.007 and INNO.0026 approved on 29 September 2021) which ensures that the analysed samples of veterinary medical centres can be used anonymously in studies and scientific research works related with this project.
Acknowledgments
The authors extend sincere gratitude to the INNO Veterinary Laboratories (Braga, Portugal) for the generous provision of results that facilitated the research conducted in this study.
References [… show]
Prvi opis osjetljivosti na protugljivična sredstva gljivica nalik kvascu u dermalnim lezijama pasa u Portugalu – retrospektivna pilot studija u trajanju od 6 godina (2018. – 2023.)
Ricardo LOPES, DVM, MSc, Department of Veterinary Sciences, University of Trás-os-Montes e Alto Douro (UTAD), Vila Real, Portugal and Department of Veterinary and Animal Sciences, University Institute of Health Sciences (IUCS), CESPU, Gandra, Portugal; Andreia GARCÊS, DVM, MSc, PhD Wildlife Rehabilitation Centre (CRAS), Veterinary Teaching Hospital, and Animal and Veterinary Research Centre (CECAV), Associate Laboratory for Animal and Veterinary Sciences (AL4AnimalS), UTAD, Vila Real, Portugal; Augusto SILVA, DVM, MSc, INNO Veterinary Laboratories, Braga, Portugal; Paula BRILHANTE-SIMÕES, DVM, MSc, Department of Veterinary and Animal Sciences, IUCS, CESPU, Gandra, Portugal, and INNO Veterinary Laboratories, Braga, Portugal; Ângela MARTINS, Department of Zootechnics, and CECAV, AL4AnimalS, UTAD, Vila Real, Portugal; Luís CARDOSO, DVM, MSc, PhD, DipEVPC, Department of Veterinary Sciences, and CECAV, AL4AnimalS, UTAD, Vila Real, Portugal; Elsa LECLERC DUARTE, DVM, MSc, PhD, DipECVM, Department of Veterinary Medicine, School of Science and Technology, and Mediterranean Institute for Agriculture, Environment and Development (MED), Global Change and Sustainability Institute (CHANGE), University of Évora, Évora, Portugal; Ana CLÁUDIA COELHO, DVM, MSc, PhD, DipECVM, Department of Veterinary Sciences, and CECAV, AL4AnimalS, UTAD, Vila Real, Portugal
Gljivične infekcije, posebice prouzročene vrstama iz rodova Malassezia i Candida, važni su doprinositelji otitisa i dermatitisa u pasa. Ova studija ispituje prevalentnost i otpornost na protugljivična sredstva različitih gljivica nalik kvascu izoliranih iz dermalnih lezija pasa u Portugalu. Od 2018. do 2023. prikupljeni su uzorci pasa s kliničkom sumnjom na gljivične infekcije i analizirani upotrebom Vitek 2 Compact® (bioMérieux, Marcyl’Étoile, Francuska) i FUNGITEST™ 60780 kompleta (Bio-Rad, Hercules, SAD). Identificirano je ukupno 24 izolata gljivica, uključujući među ostalima Malassezia pachydermatis, Candida albicans. Ispitivanje osjetljivosti otkrilo je veliku osjetljivost na protugljivična sredstva poput flukonazola, ketokonazola, itrakonazola i mikonazola za većinu vrsta, sa značajnom otpornošću zamijećenom kod Rhodotorula mucilaginosa na više protugljivičnih sredstava. Srednje razine otpornosti su otkrivene u vrste Malassezia na flucitozin i nekoliko drugih protugljivičnih sredstava. Ovi nalazi naglašavaju potrebu za kontinuiranim nadzorom i razboritom uporabom protugljivičnih sredstava da bi se spriječio razvoj otpornosti. Studija naglašava i važnost ponovnog razmatranja protokola liječenja za infekcije Malasseziom u pasa, obzirom na zamijećene obrasce otpornosti. Preporučuje se dodatno istraživanje za ispitivanje genetskih i molekularnih mehanizama koji su inherentni za otpornost na protugljivična sredstva i razvoj novih terapeutskih agensa.
Ključne riječi: otpornost na protugljivična sredstva, klinička patologija, dermatologija, gljivice, infekcije, mikologija, jedno zdravlje, planetarno zdravlje, gljivice nalik kvascu

