Mastitis pathogens in goats and sheep in the Republic of Croatia and their sensitivity to antimicrobials

L. Cvetnić*, M. Samardžija, T. Sukalić, D. Lukačević, M. Lolić, G. Kompes, B. Habrun, A. Bagarić, Ž. Cvetnić and M. Benić
Abstract
This paper outlines the results of researching the incidence of mastitis in small ruminants in the Republic of Croatia over a six-year period (2017 to 2022). During this period, microbiological testing was performed on 801 samples of udder secretions from small ruminants in 13 counties and the city of Zagreb, with 178 samples (22.2%) testing positive for mastitis pathogens. Of the total 609 udder secretion samples from goats, mastitis agents were isolated from 150 samples (24.6%), while the 192 udder secretion samples from sheep revealed 28 (14.6%) contained mastitis agents. The highest prevalence of mastitis pathogens in goat udder secretions was found in 2018 (31.8%), and in sheep in 2020 (32.9%). Microbiological testing identified 16 species and genera of bacteria. The most common species were Staphylococcus aureus, isolated from 96 (53.9%) samples, Staphylococcus spp. (46 samples; 25.8%), Streptococcus spp. (7; 3.9%), Klebsiella spp. and Corynebacterium spp. (each 5; 2.8%), Escherichia coli (4; 2.2%), Streptococcus uberis and Proteus spp. (each 3; 1.7%), and Pseudomonas spp. (2; 1.1%). Other species and genera, such as Streptococcus agalactiae, Streptococcus dysgalactiae, Pasteurella spp., Mannheimia haemolytica, Enterococcus spp., Enterobacter and Serratia spp. were isolated sporadically, each from one sample. The isolated bacterial cultures were tested for sensitivity to antimicrobials using the disc-diffusion method to amoxicillin with clavulanic acid, ampicillin, cefaperazone, enrofloxacin, kanamycin, cloxacillin, lincomycin, neomycin, novobiocin, penicillin, streptomycin, sulfamethoxazole with trimethoprim, and tetracycline. Sensitivity of the isolated agents to the antimicrobials was most pronounced towards cefoperazone and enrofloxacin, regardless of the species or genera of the isolated mastitis agent. The odds of isolating a mastitis agent from goat udder secretions was 1.61 times higher than for sheep (P<0.05). This study confirmed that the occurrence of mastitis in small ruminants in Croatia is comparable to incidences reported in other Mediterranean countries.
Key words: mastitis; prevalence; antimicrobial resistance; small ruminants
Introduction
Mastitis is an inflammation of the mammary gland, regardless of the cause (Radostits et al., 2000). The host, pathogen, and predisposing environmental factors all play an important role in the development of mastitis.
Most cases of mastitis in small ruminants occur as a result of bacterial infection of the udder, due to the transfer of bacterial pathogens via milk or milking equipment during milking, or from the introduction of bacteria from the animal’s skin or the environment. Mastitis can occur in two forms, as subclinical or clinical mastitis, with various clinical manifestations, from short-term acute infections to chronic infections that are resilient and long-lasting (Mwenge Kahinda, 2021). The most common form of mastitis in sheep and goats is subclinical mastitis, with a prevalence from 5 to 30%, and occasionally to 50% (Contreras et al., 2007). Clinical mastitis in small ruminants is usually seen with an incidence of less than 5%, and most often occurs at the start of machine milking and during the first third of lactation. These properties are distinctive in small ruminants in comparison with dairy cows (Bergonier et al., 2003). Not addressing an acute udder infection leads to the development of chronic forms of mastitis, reduced quantity and quality of milk, loss of udder function, and ultimately early removal of affected sheep or goats, and sometimes in death (Arsenault et al., 2008; Gonzalo et al., 2002; Barron-Bravo et al., 2013; De Olives et al., 2013). Additionally, the reduced milk production affects other forms of productivity, as it results in lower weight gain of suckling lambs (Arsenault et al., 2008). More than 30 bacterial species have been isolated from sheep and goats with mastitis (Arsenault et al., 2008). The most common microorganisms isolated in udder infections of small ruminants are Staphylococcus (S.) aureus, Staphylococcus spp, Streptococcus spp. and Mannheimia spp. (Morg et al., 2007; Marogna et. al., 2010; Smith et. al., 2015). Bergonier et. al., (2003) in France reported that infections caused by members of the genus Staphylococcus spp., except S. aureus, most often ranged from 25-93%, infections caused by S. aureus from 3 to 37%, and these pathogens were isolated from clinically mild cases of chronic udder infections in small ruminants. Spuria et al. (2017) in Italy proved a high percentage (71.9%) of various bacterial species, ecologically opportunistic bacteria and other microorganisms from the tissues of macroscopically health udders taken during the regular slaughter of sheep and goats. The authors stated that the most mastitis in goats and sheep is associated with S. aureus. Achek et al. (2019) stated that the most common mastitis pathogens in sheep in Algeria were S. aureus and coagulase-negative staphylococci.
Lafi et al. (1998) in Jordan reported the highest isolation of coagulase-negative staphylococci in 17.8%, E. coli in 13.6%, and S. aureus in 6.8% of the flock. Langoni et al. (2006) in Brazil reported an incidence of mastitis in goats caused by the species Staphylococcus epidermis in 50% of tested samples, Streptococcus agalactiae in 13.6%, Staphylococcus aureus in 11.4%, with other species appearing more rarely. In Croatia, mastitis in small ruminants has rarely been described. Cvetnić et al. (2016) reported that pathogens were isolated from 58 samples (15.5%) of 375 samples of goat udder secretions, and that the causative agent in 27 of these samples (46.6%) was S. aureus.
The aim of this paper was to describe the distribution of mastitis in sheep and goats in the Republic of Croatia in the period from 2017 to 2022, determine the incidence of different mastitis pathogens and their sensitivity to antimicrobials used in the treatment of mastitis.
Materials and methods
Milk sampling
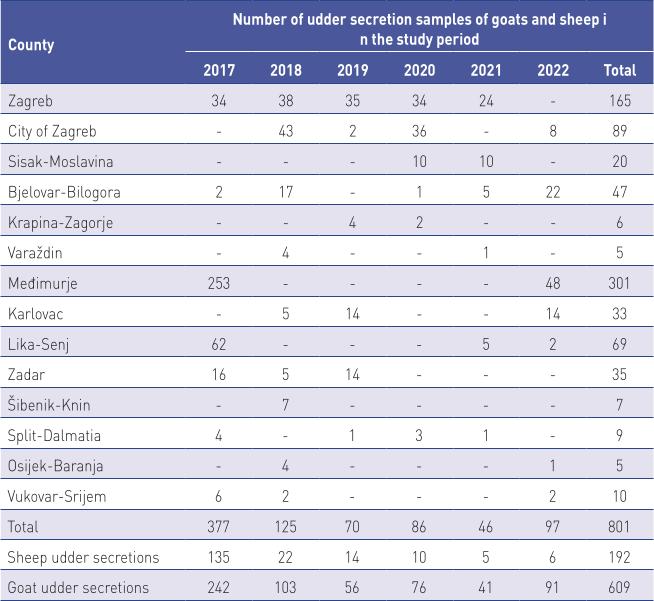
In the period from 2017 to 2022, a total of 801 samples of udder secretions of small ruminants collected from 14 counties in Croatia were delivered to the laboratory, of which 609 samples were goat udder secretions and 192 samples were sheep udder secretions.
Sheep and goat udder secretion samples for microbiological testing were collected at sheep and goat farms, and delivered to the Croatian Veterinary Institute Zagreb and Veterinary Institutes in Križevci, Vinkovci and Split.
Samples were transported on ice and kept at +4˚C until analysis, which was performed in the laboratory within 12 hours of receipt (Table 1).
Laboratory analysis
Microbiology testing. Microbiology testing was performed in compliance with the generally accepted recommendations listed in the Laboratory handbook on bovine mastitis (National Mastitis Council, 2017).
From samples of each udder half, 0.01 mL of the sample was inoculated using a single-use 10 mm inoculation loop onto the surface of a Petri dish containing esculin blood agar.
The inoculated dishes were incubated at 37°C for 24 hours, and then controlled for colony growth on the agar surface. The remainder of the sample was kept in the refrigerator at 4°C until the end of analysis. For samples that showed no microbial growth after 24-hour incubation, 0.1 mL of the sample was again inoculated onto half the surface of the Petri dish, with control of growth performed after another 24 hours. After incubation, bacterial colonies on the inoculated agar were determined. Morphological (colony shape, size and structure) and physiological (formation of pigment, CAMP phenomena, breakdown of esculin, ability to clot rabbit plasma, Gram staining) traits were considered in the determination.
All isolated bacteria were identified using the Micronaut system. A suspension of isolated bacteria grown in pure culture was inoculated into the wells of a microtitration plate with added substrates. After incubation of the inoculated microtitration plates, the results were read using spectrophotometry. The computer program reads and processes the results, and compiles the final identification report.
The sensitivity of the researched strains of Staphylococcus aureus to antimicrobial drugs was determined using the disc diffusion procedure.
To determine sensitivity, a suspension of selected colonies grown on blood or nutrient agar was prepared in saline solution to a density of 0.5 according to McFarland. Optical density of the suspension was set using the Densimat device (BioMerieux), according to the standard of the Clinical and Laboratory Standards Institute (CLSI, 2015). After preparing the suspension to the appropriate density, a sterile cotton swab was used to layer the suspension onto a Petri dish three times in different directions, by turning the dish by 60° between each layer to cover the entire surface of the agar. Using a dispenser, we placed discs on the agar surface, left the dish for 15 minutes at room temperature and then placed in the thermostat at 35°C. Müller-Hinton agar (Merck, Germany) was used for the procedure. Results were read after 16–20 hours of incubation. We measured the diameter of zones of complete growth inhibition. Strains were assessed as susceptible (S), intermediate (I) or resistant (R) (CLSI, 2015).
The degree of sensitivity was determined using discs with known concentration: amoxicillin with clavulanic acid AMC (20/10 µg), ampicillin AMP (25 µg), cefaperazone CFP (75 µg), enrofloxacin ENR (5 µg), kanamycin KAN (30 µg), cloxacillin CLX (5 µg), lincomycin LIN (30 µg), neomycin NE (30 µg), novobiocin NB (30 µg), penicillin P (10 µg), streptomycin S (10 µg), sulfamethoxaloze/trimethoprim SXT (23/1 µg), tetracycline TC (30 µg), all by the manufacturer Becton Dickinson.
Data analysis was performed using the program Stata 18.0 (Stata corp, USA).
The number of samples was shown by geographical origin and year of sampling.
Sensitivity/resistance to antimicrobials was reported descriptively. The association between infection prevalence and animal species and year of sampling was tested using the chi-square test, and the results of statistical analysis expressed using p values. Statistical significance was set at a value of p<0.05.
Animal species and year of sampling were analysed in the logistic regression model, and the results reported as the odds ratio for isolating the causative agent from the udder secretions (Odds ratio, OR).
Results
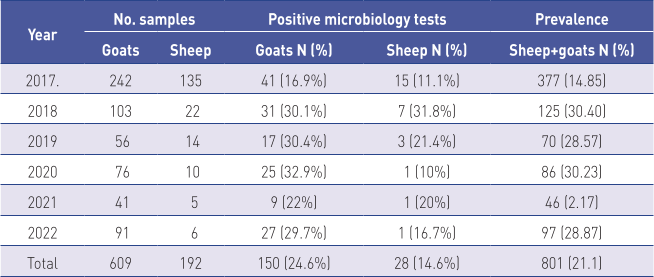
During the 6-year study period (2017 to 2022), a total of 801 udder secretion samples were collected from small ruminants from 13 counties and the city of Zagreb. Samples were tested microbiologically and mastitis pathogens were isolated from 178 (21.2%) of samples. Of the 609 goat udder secretion samples, positive results were detected in 150 samples (24.6%), as opposed to 28 of 192 samples (14.6%) of sheep udder secretion samples. The observed difference in the incidence of isolation of pathogens between goats and sheep was statistically significant (p<0.05). The prevalence of mammary gland infections in sheep and goats was statistically different based on year of sampling, ranging from 2.17% (in 2021) to 30.40% (2018) (P<0.001) (Table 2).
In the logistic regression model, we analysed the variables of animal species and year of sampling, as both variables were statistically significantly associated with the outcome of microbiological testing, i.e., with the isolation of causative agents in the chi-square test. The results of the logistic regression indicate that the outcome of the testing, i.e., the odds of isolating the pathogen from udder secretions are statistically significantly associated with the animal species. The odds for isolating the mastitis pathogen from goat udder secretions were 1.61 times higher than from sheep’s milk (P=0.04) (Table 3).
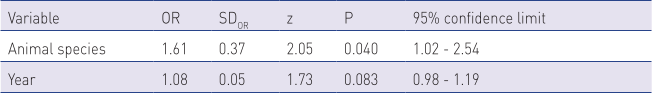
OR – odds ratio; SDOR – standard deviation; z – z value; P – p-value
In total, 16 species or genera of bacteria were isolated from the small ruminant udder secretions. The most commonly isolated were the species Staphylococcus (S.) aureus from 96 (53.9%) samples, Staphylococcus spp. (25 samples; 14.0%), Staphylococcus spp. (CNS) (21; 11.8%), Streptococcus spp. (7; 3.9%), Klebsiella spp. and Corynebacterium spp. (each 5; 2.8%), Escherichia coli (4; 2.2%), Streptococcus uberis and Proteus spp. (each 3; 1.7%), Pseudomonas spp. (2; 1.1%), while other species such as Streptococcus agalactiae, Streptococcus dysgalactiae, Pasteurella spp., Mannheimia haemolytica, Enterococcus spp., Enterobacter and Serratia spp. were each isolated from only one small ruminant udder secretion sample. The most frequently isolated species in sheep were S. aureus (18; 64.3%), Staphycoccus spp. (CNS) (8; 28.6%) and Proteus spp. (2; 7.1%) of the 28 bacterial species isolated from sheep udder secretions (Table 4).

The isolated bacterial cultures were tested for antimicrobial sensitivity and the results are shown in Table 5.
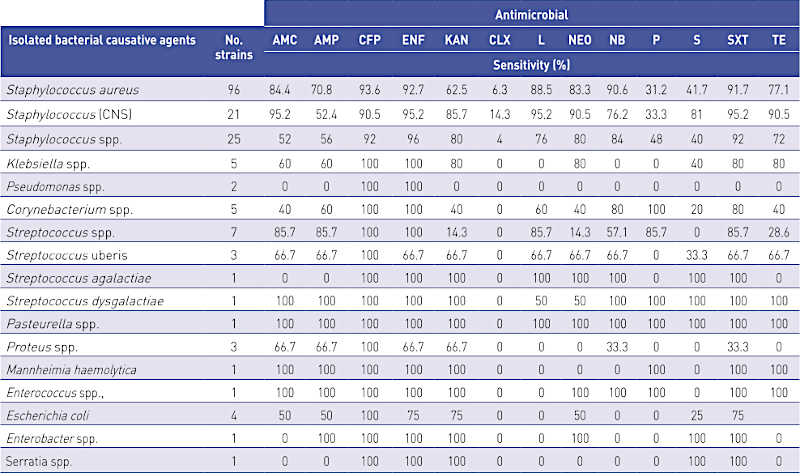
AMC- amoxicillin; AMP-ampicillin; CFP-cefoperazone; ENF-enrofloxacin; KAN-kanamycin; CLX – cloxacillin; L-lincomycin; NEO-neomycin; NB-novobiocin; P-penicillin; S-streptomycin; SXT-sulfamethoxazole + trimethoprim; TE-tetracycline.
Discussion
Inflammation of the mammary gland is a complex disease caused primarily by microorganisms. Its prevalence and incidence are also affected by the environment, resilience of the host, and farm management practices.
The causative agents are primarily various gram-positive and gram-negative bacteria and fungi (Deb et al., 2013). Udder infections cause significant damages to the dairy industry due to reduced quantity and quality of milk production (Benić, 2001). The occurrence of mastitis is also due to the non-abidance of fundamental zoohygiene and zootechnical measures and of the fundamental postulates of hygienic milking, and poor functional or technical state of milking machines (Benić, 2011). The aetiology of mastitis has many causes and it cannot be realistically expected that the management of mastitis will ever be completely resolved (Cergolj, 2003). Though in recent decades more than 95% of dairy products around the world are made of cow’s milk, in Mediterranean countries, sheep’s and goat’s milk are particularly important as they are part of the cultural heritage (Michaelidou, 2008; Rupić, 2010). Milk products of small ruminants account for a significant share of the economy in many countries, particularly in the Mediterranean and Middle East (Park and Haenlein, 2006; Akers and Nickerson, 2011).
During the 6-year study, the presence of the mastitis pathogens was confirmed in 178 (22.2%) of 801 samples of small ruminant udder secretions. The most commonly isolated species were S. aureus from 96 samples (53.9%), Staphylococcus spp. (25; 14%) and Staphylococcus spp. (CNS) (21; 11.8%) while all other species were found less often. Bergonier et al. (2003) stated that chronic infectious mastitis appears more often in small ruminants, while staphylococci are the main aetiological cause of mastitis in goats. Koop et al. (2012) stated that S. aureus is the causative agent of primary interest in dairy goats.
Cvetnić et al. (2016) described similar results in Croatia during 2015, when 58 of 375 (15.5%) udder secretion samples contained pathogens.
Microbiology testing confirmed that the most common species in that study were S. aureus (46.6%), Streptococcus dysgalactiae (22.4%), Staphylococcus spp. (CNS) (20.7%), Enterococcus spp. (6.9%) and E. coli (3.4%).
During routine checks for mastitis, several flocks of goats in northern Italy showed a prevalence of 40.2% among 305 tested samples of udder secretions. Bacteriology testing found that Staphylococcus spp. (CNS) was most common (80% samples), S. aureus (6%), while other environmental microorganisms were found in 14% of samples, and the most common of these was Enterococcus faecalis (Moroni et al., 2005). S. aureus was one of the most commonly confirmed pathogenic microorganisms in udder secretions of goats and sheep in Switzerland, with 31.7% of goat’s milk samples and 33.3% of sheep’s milk samples testing positive (Marogna et al., 2012; Muehlherr et al., 2003). In Italy, Dore et al. (2016) found coagulase-negative staphylococci in 48.89% of sheep flocks and 54.2% of goat flocks. S. aureus was detected in 39.8% of sheep samples and 41.5% of goat samples, followed by Streptococcus uberis (11.8% in sheep, 5.9% in goats), Enterobacteriaceae (15% in sheep, 16.2% in goats), while other pathogens were isolated in smaller frequencies. Research on 12 flocks of dairy sheep in Greece found a 51.7% incidence of subclinical mastitis, which varied from 25 to 75% at the farm level. The most frequent cause of mastitis was infection by various species of staphylococci, most commonly S. aureus (19.2%) (Charalambia et al., 2023). Las Helas et al. (1999) examined 17 flocks of sheep in the Madrid region of Spain and found a flock-level prevalence from 4.5 to 67%. Coagulase-negative staphylococci were most commonly represented (67% of isolates) and members of the genus Corynebacterium were the second most identified group, with 10% of samples from 415 flocks testing positive.
In southern Turkey, Ergun et. al. (2009) confirmed a prevalence of subclinical mastitis in individual flocks from 1.9 to 21.9%, with members of the genus Staphylococcus spp. (CNS) most common, accounting for 76.5% of isolates. The authors concluded that subclinical mastitis was not widely distributed and did not pose a significant health threat in southern Turkey. Lafi et al. (1998) conducted a study of 46 flocks of sheep to determine the spread of mastitis in Jordan. The most common causes of subclinical mastitis in sheep were coagulase-negative staphylococci (17.8%), E. coli (13.6%), Streptococcus agalactiae (6.8%), and Staphylococcus agalactiae (6.8%). From clinical cases of mastitis, the most commonly isolated species were S. aureus (22%) and E. coli (14,2%).
Langoni et al. (2014) studied subclinical mastitis in goats in the Sao Paulo region of Brazil and found that the most common species were Staphylococcus epidermidis (50%), Streptococcus agalactiae (13.6%), Staphylococcus aureus (11.4%) and Corynebacterium bovis (8.6%). Other microorganisms were also isolated, such as Candida albicans, Bacillus spp., Pasteurella multocida, Escherichia coli and Acinetobacter spp.
McDougall et al. (2014) confirmed the presence of mastitis pathogens in 23.3% of goat udder secretions in New Zealand, and the most common species isolated were coagulase-negative staphylococci (CNS) (13.4%) and Corynebacterium spp. (7.3%).
Zhao et al. (2015) reported causative agents of mastitis in 45.8% of goats in a study in China.
The most commonly isolated species were coagulase-negative staphylococci (59.5%), S. aureus (15.2%), E. coli (11.4%) and Streptococcus spp. (10.9% of samples).
Conclusion
The present study showed that the prevalence of mastitis in small ruminants in Croatia is comparable to the prevalence in other Mediterranean countries. The dominant cause of mammary gland infections in both sheep and goats is S. aureus, followed by coagulase-negative staphylococci. The isolation of pathogens from udder secretion samples, and determination of the sensitivity of these agents to antimicrobials, is the basis for controlling mastitis in small ruminants.
It is evident that different species of isolated bacteria require different treatment approaches, from the selection of antibiotics and treatment methods. Due to the general lack of specifically designed treatments for small ruminants, most often treatment methods are the same or similar to those used in cows. Milking hygiene is key for success of the mastitis control programme in small ruminants. Procedures aimed at preventing mastitis include a number of factors, such as: selection of antimicrobials, sensitivity of the microorganism, duration of treatment, and drug dose. Excessive and uncontrolled use of antibiotics increases the risk of developing bacterial resistance to frequently used antimicrobials, which has become a public health issue. In recent years, members of the genus Staphylococcus are frequently mentioned in relation to antimicrobial resistance, and this group was also the most commonly isolated group of bacteria in this study. In comparison with other groups of pathogenic bacteria, clinical isolates of coagulase-positive and coagulase-negative staphylococci isolated from humans and animals are increasingly displaying higher resistance to beta-lactam antibiotics, including penicillin and ampicillin.
References [… show]
Uzročnici mastitisa koza i ovaca u Republici Hrvatskoj i njihova osjetljivost prema antimikrobnim tvarima
Dr. sc. Luka CVETNIĆ, znanstveni suradnik, Hrvatski veterinarski institut, Zagreb, Hrvatska; dr. sc. Marko SAMARDŽIJA, redoviti profesor, Veterinarski fakultet Sveučilišta u Zagrebu, Hrvatska; dr. sc. Tomislav SUKALIĆ, znanstveni suradnik, Hrvatski veterinarski institut – podružnica Veterinarski institut Križevci, Hrvatska; dr. sc. Damir LUKAČEVIĆ, stručni suradnik, Hrvatski veterinarski institut – podružnica Veterinarski institut Split, Hrvatska; dr. sc. Marica LOLIĆ, stručna savjetnica, Hrvatski veterinarski institut – podružnica Veterinarski institut Vinkovci, Hrvatska; dr. sc. Gordan KOMPES, znanstveni savjetnik, dr. sc. Boris HABRUN, znanstveni savjetnik u trajnom izboru, redoviti profesor, Antonela BAGARIĆ, dr. med. vet., Hrvatski veterinarski institut, Zagreb, Hrvatska; akademik Željko CVETNIĆ, Hrvatski veterinarski institut – podružnica Veterinarski institut Križevci, Hrvatska; dr. sc. Miroslav BENIĆ, znanstveni savjetnik u trajnom zvanju, izvanredni profesor, Hrvatski veterinarski institut, Zagreb, Hrvatska
U radu su opisani rezultati istraživanja učestalosti mastitisa u malih preživača u Republici Hrvatskoj u razdoblju od šest godina (2017. do 2022.). Tijekom istraživanog razdoblja na području trinaest županija i Grada Zagreba mikrobiološkim pretragama pretražen je 801 uzorak sekreta vimena malih preživača, a uzročnici mastitisa izdvojeni su iz 178 uzoraka (22,2 %). Od pretraženih 609 uzoraka sekreta vimena koza, uzročnici mastitisa izdvojeni su iz 150 uzoraka (24,6 %) sekreta, dok je od 192 pretražena sekreta vimena ovaca iz 28 (14,6 %) izdvojen neki od uzročnika mastitisa. Najviša prevalencija uzročnika mastitisa u sekretu vimena koza utvrđena je 2018. godine (31,8 %), a u sekretu vimena ovaca 2020. godine (32,9 %). Mikrobiološkom pretragom izdvojeno je 16 različitih vrsta i rodova bakterija. Najčešće je vrsta Staphylococcus aureus, izdvojena iz 96 (53,9 %) uzoraka, Staphylococcus spp. iz 46 (25,8 %), Streptococcus spp. iz 7 (3,9 %), Klebsiella spp. i Corynebacterium spp. iz 5 (2,8 %), Escherichia coli iz 4 (2,2 %), Streptococcus uberis i Proteus spp. iz 3 (1,7 %) te Pseudomonas spp. iz 2 (1,1 %) uzorka. Ostale vrste i rodovi poput Streptococcus agalactiae, Streptococcus dysgalactiae, Pasteurella spp., Mannheimia haemolytica, Enterococcus spp., Enterobacter i Serratia spp. izdvojene su sporadično iz po jednog uzorka. Izdvojene bakterijske kulture podvrgnute su testu osjetljivosti na antimikrobne tvari disk-difuzijskom metodom prema amoksicilinu s klavulanskom kiselinom, ampicilinu, cefaperazonu, enrofloksacinu, kanamicinu, kloksacilinu, linkomicinu, neomicinu, novobiocinu, penicilinu, streptomicinu, sulfametoksazolu s trimetoprimom i tetraciklinu. Osjetljivost izdvojenih uzročnika prema antimikrobnim tvarima najizraženija je prema cefoperazonu i enrofloksacinu, neovisno o vrsti ili rodu izdvojenog uzročnika mastitisa. Izgledi za izdvajanje uzročnika mastitisa iz sekreta vimena koza 1,61 puta su veći od izgleda u ovaca (P<0.05). Našim istraživanjem utvrdili smo da je u Republici Hrvatskoj učestalost pojave mastitisa u malih preživača usporediva s učestalošću zabilježenom u drugim mediteranskim zemljama.
Ključne riječi: mastitis, prevalencija, antimikrobna rezistencija, mali preživači

